Postoperative recurrent Crohn’s disease is common and often clinically silent at onset, requiring objective assessments for diagnosis and surveillance. Patients with a history of multiple bowel resections, penetrating disease, or who smoke cigarettes after surgery are at highest risk for disease recurrence. Antibiotics, aminosalicylates, and immunomodulators modestly reduce the risk of clinical disease recurrence. In contrast, monoclonal antibodies, specifically anti-tumor necrosis factor (TNF) medications as well as the anti-integrin agent vedolizumab, are effective at suppressing disease recurrence and may have the potential to alter the natural course of disease after surgery. In this manuscript, the management of postoperative Crohn’s disease is summarized, and a simplified approach to prevention, monitoring, and treatment is provided.
Risk and Diagnosis of Postoperative Crohn’s Disease
Despite significant medical therapeutic advances, as many as 20-30% Crohn’s disease (CD) patients require bowel surgery, mostly commonly for stricturing or penetrating complications, e.g., fistula, intraabdominal abscess. Surgery is not curative of CD, and postoperative recurrence (POR) of CD occurs in the majority of patients. In the pre-biologic era (circa 2000), natural history studies found that 70–90% of CD patients developed endoscopic evidence of disease activity within 1 year of their surgery, and that 30–60% of postoperative CD patients became symptomatic from recurrent disease within 3–5 years of their surgery. Consequently, up to 50% of postoperative CD patients in the pre-biologic era required repeat surgery within 5 years of their first surgery.
Postoperative CD recurrence is often clinically silent with only 1/3 of patients reporting symptoms of CD despite endoscopic disease activity in the majority postoperatively. Thus, relying on symptoms postoperatively significantly underestimates mucosal disease activity and may miss an opportunity for disease interception. Fortunately, with increased utilization of postoperative management strategies, more modern estimates suggest the risk of recurrence is diminishing.
The degree of endoscopic disease activity correlates with subsequent progression to symptomatic recurrence. Thus, ileocolonoscopy is the current gold standard for POR assessment and is recommended to be performed within 6-12 months postoperatively. More delayed endoscopic assessments are associated with worse clinical outcomes. Disease activity is traditionally estimated using the Rutgeerts score, which defines activity on a 0 (normal) to 4 (severe) scale based on the extent of aphthous ulcerations in the neoterminal ileum. The more severe the endoscopic recurrence, e.g. i3 or i4, the more likely the development of clinical symptoms, i.e. clinical recurrence, and requirement for future surgery, i.e. surgical recurrence.
Table 1. Summary of Gastroenterological Society Definitions of High-Risk Features for Postoperative Crohn’s Disease Recurrence.
| American College of Gastroenterology (ACG) | American Gastroenterological Association (AGA) | European Crohn’s and Colitis Organization (ECCO) | British Society of Gastroenterology (BSG) |
| Classified as high risk with ≥ 1: • Active tobacco use (especially in women and heavy smokers) • ≥2 prior surgeries • presence of penetrating disease* | Classified as high risk with ≥ 1: • Active tobacco use • ≥2 prior surgeries for penetrating disease, with or without perianal disease • Age <30 years | Classified as high risk with ≥ 1: • Smoking • ≥ 1 prior intestinal surgery • Penetrating disease at index surgery • Perianal location • Granulomas or myenteric plexitis in • resection specimen • Extensive small bowel resection (>50 cm) 2023 ECCO workshop includes: • Immune-mediated inflammatory diseases • Extra-intestinal manifestations • Prior colon involvement | Classified as high risk if actively smoking or with ≥ 2: • Multiple resections • Preoperative penetrating disease behavior Perianal disease • Granulomas in resection specimen • Myenteric plexitis in the proximal resection margin • Extensive bowel disease (>50 cm) |
Though ileocolonoscopy is sensitive at detecting POR, the invasive nature of the test is associated with patient discomfort, high cost, and procedural risk. Thus, noninvasive assessments are of particular interest. Fecal calprotectin (FCP) levels, produced by gastrointestinal leukocytes and epithelial cells at sites of mucosal injury and inflammation, correlate with endoscopic recurrence while normal levels have a strong negative predictive value. Based on available data, FCP cutoffs between 100-150 ug/g have been proposed, identifying endoscopic recurrence with 70-89% sensitivity, 58-69% specificity, and negative predictive values > 90%. Consequently, the American Gastroenterological Association (AGA) now advocates for fecal calprotectin-guided surveillance strategies as a cost-effective alternative to universal endoscopic evaluation in lower-risk patients within the first year postoperatively.1 AGA guidelines recommend assessing FCP between 6 and 12 months postoperatively. Earlier assessments are being explored.
Imaging modalities including computed tomography (CT) and magnetic resonance (MR) have both shown acceptable correlation to endoscopic disease activity, though they have disadvantages of cost, poorly tolerated oral contrast, radiation (CT), and limited availability (MR). Intestinal ultrasound (IUS) has emerged as a promising radiation-free, cost-effective point-of-care imaging modality capable of detecting POR with comparable accuracy to ileocolonoscopy in expert hands.2 Combining FCP assessments with IUS can further improve test characteristics. Consequently, postoperative monitoring algorithms are increasingly incorporating these non-invasive tools when available.
Risk Factors for Postoperative Recurrence
Factors associated with POR include clinical, disease, surgical, histologic, microbiotic, and molecular characteristics. Active smoking after surgery doubles the risk of endoscopic, clinical, and surgical recurrence and smoking cessation can reduce recurrence rates. Younger age at disease onset and rapid progression (<10 years) to surgical resection may increase recurrence risk. A history of prior surgical resections for Crohn’s may impart the strongest risk for future POR.3,4 Penetrating disease behavior (fistula, abscess) at the time of surgery is associated with increased clinical and surgical recurrence.
Surgical approach and anastomosis technique may influence POR. These include more extensive surgical resections of the mesentery as well as novel anastomotic orientations such as the Kono-S configuration. Such approaches are being explored in randomized trials. Furthermore, histologic findings in the resection specimen including presence of granulomas, myenteric and submucosal plexitis, and positive surgical margins may identify individuals at increased risk for POR.5,6
Table 2. Efficacy of various therapies and knowledge gaps for the prevention of postoperative Crohn’s disease recurrence.
| Therapy/Intervention | Postoperative Recurrence Prevention Relative Efficacy |
| Curcumin | – |
| Vitamin D | – |
| Enteral Nutrition | + |
| Probiotics | – |
| Nitroimidazole/Antibiotics | + |
| Mesalamine | – |
| Budesonide | – |
| Thiopurines | + |
| Anti-TNF | +++ |
| Vedolizumab | ++ |
| Ustekinumab | + |
| Anti-IL-23s | ? |
| Janus kinase inhibitors | ? |
Microbiome, serologic, genetic, and other “-omics” signatures have been described in individuals who progress to POR, but data remains inconclusive for routine clinical care at the current time.
Risk stratification has been adopted in various gastroenterological societal guidelines (Table 1). Generally, patients at high risk for recurrence include those who are actively smoking, multiple prior surgical resections, and penetrating disease behavior, with or without perianal disease. Such risk stratification may help identify patients warranting more aggressive treatment and monitoring after surgery.
Medical Prophylaxis for Preventing Postoperative Crohn’s Disease
Conventional medical therapies including antibiotics (e.g. metronidazole), aminosalicylates, and immunomodulators modestly reduce the risk of clinical disease recurrence and may be considered particularly in resource-limited settings; however, comparative analyses suggest that more novel targeted therapies are more efficacious for this purpose (Table 2).7
Anti-tumor necrosis factor (TNF) therapy postoperatively prevents endoscopic POR and may have the potential to change the natural course of Crohn’s disease after surgery. The seminal PREVENT trial demonstrated that infliximab when used prophylactically (within 2-4 weeks postoperatively) in individuals at high risk for recurrence significantly reduced endoscopic recurrence at week 76 compared to placebo (22.4% vs. 51.3%, P < 0.001), although not clinical recurrence (12.9% vs 20.0%, P=0.097).8 This protective effect appears to extend to other anti-TNFs as adalimumab has also been found to prevent POR in several studies.9-11
In addition to anti-TNFs, the anti-integrin molecule vedolizumab (VDZ) has been recently assessed for postoperative prevention. The REPREVIO randomized trial demonstrated that CD patients with at least one POR risk factor assigned to receive VDZ within 4 weeks of ICR had a 77.8% probability of having a significantly lower modified Rutgeerts score and lower proportion with severe endoscopic recurrence (mRS ≥i2b) than the placebo group.12 Rates of severe endoscopic recurrence were 23.3% and 62.2% for the VDZ and placebo groups respectively. Notably, 62.8% and 62.2% of the VDZ group and placebo group respectively were anti-TNF experienced. These findings suggest that VDZ can be an efficacious preventative strategy either in those who are anti-TNF experienced or as an initial postoperative therapy. No prospective studies have evaluated comparative efficacy between anti-TNFs and VDZ.
Data is emerging on the effectiveness and comparative efficacy of newer biologics or oral small molecules. These include the anti-interleukin 12/23 agent ustekinumab, anti-interleukin 23 molecules, and Janus kinase inhibitors. While these molecules are often utilized clinically for the purpose of preventing recurrence, data supporting efficacy in the postoperative setting is currently lacking.
Watch and Wait: Endoscopically-Guided Postoperative Management
Given that most, but not all, patients will develop postoperative recurrent disease suggests that universal prophylactic biologic therapy in all postoperative Crohn’s disease patients would mean overtreating a subset with consequent risks and costs. An alternative strategy would be to objectively monitor individuals postoperatively and if disease activity is identified and confirmed endoscopically, then to initiative or adjust treatment at that time. Such endoscopically-guided detection and treatment of POR was assessed in the pivotal POCER study.13 The authors demonstrated that colonoscopy at 6 months after surgery with treatment escalation for identified recurrence improved endoscopic rates at 18 months compared to routine care without a 6 month colonoscopy (49% vs. 67%, P = 0.03). Furthermore, a multicenter, retrospective cohort study comparing medical prophylaxis vs. endoscopically-guided strategies found that while there was a significant reduction in 12-month endoscopic POR in high-risk patients on medical prophylaxis when compared to the endoscopy-driven group (24.3% vs 44.5%, p=0.03), there was no significant difference in time to clinical POR for both low- and high-risk groups over the 3 year study follow up period.14 However, other retrospective studies have suggested that medical prophylaxis was associated with a significantly lower risk of endoscopic POR.15
Strategies for Postoperative Crohn’s Disease Management
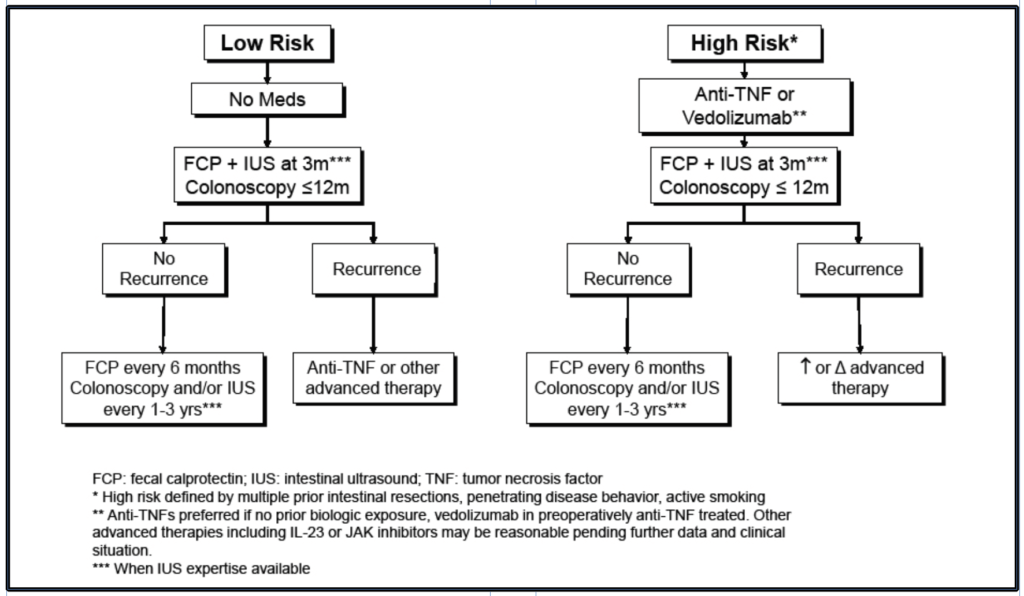
Consequently, key questions that remain in the practical management of postoperative Crohn’s disease are: (1) which patients should receive immediate postoperative therapy as prophylaxis against POR, and (2) which patients would it be reasonable to wait to treat endoscopic recurrence? Until evidence more clearly determines the optimal strategy for individual patients, the current prevailing approach for postoperative Crohn’s disease management is to stratify postoperative treatment based on risk and treat those patients at high risk for recurrence with prophylactic medical therapy (Figure 1). High risk factors include active smoking, multiple prior Crohn’s-related surgeries, penetrating disease behavior (e.g., intraabdominal fistula or abscess). The authors also consider those with residual disease (gross or positive margins) after surgery to be at high risk for POR. High-risk individuals should be considered for initiation of prophylactic biologic therapy within 4 weeks of surgery. Such risk-stratified utilization of medical prophylaxis has been shown to reduce rates; however, it should be noted that when utilized in low-risk patients, prophylaxis was similarly effective.14,16
For individuals at high risk on medical prophylaxis, or with surgical or histopathologic factors for recurrence, e.g., myenteric plexitis, transmural lesions, granulomas all requiring validation studies, one can consider incorporating early biomarker monitoring with FCP as well as IUS at 3 months postop. If FCP is elevated > 150 ug/ml or IUS features of disease activity, earlier colonoscopy (prior to month 6) to assess disease activity and adjust treatment regimen is reasonable. If these early assessments are normal or minimal, a gold standard colonoscopy between 6-12 months should be used to guide therapy and follow up decisions. Concurrent FCP assessment (measured prior to colonoscopy preparation) is helpful if future biomarker monitoring is desired to align FCP levels to contemporaneous endoscopy findings.
In high-risk patients who are receiving preoperative biologic therapy and plan to utilize biologic therapy postoperatively, it is important to distinguish preoperative therapeutic failure (e.g., active disease progression despite adequate drug exposure) from “failure” due to preexisting damage (e.g., fibrostenotic stricture) or complication (e.g., penetrating disease). With verified therapeutic failure, the biologic mechanism of action should be changed postoperatively. It is the authors opinion that with a preexisting stricture or complication, the preoperative biologic exposure does not necessarily represent a true therapeutic failure but was rather instituted too late in the disease course to reverse the existing tissue damage. Consequently, the agent or therapeutic class may be continued postoperatively for prophylaxis, particularly for anti-TNFs (+/- immunomodulator). Despite historical concerns about risk of perioperative complications with biologics, more recent large prospective studies controlling for confounding factors (e.g., malnutrition, steroids) have not seen a detrimental effect of perioperative biologic exposure.17 Thus, in this situation, the authors also frequently continue the biologic dosing throughout the perioperative period after discussing with the surgical team.
Low-risk patients are identified by those without prior surgical history, nonsmokers, and lacking other high-risk factors. Individuals identified as low risk for POR could refrain from prophylactic biologic therapy and instead be monitored with FCP and IUS at 3 months with subsequent colonoscopy within 12 months of surgery with treatment decisions guided by endoscopic findings.
Importantly, early endoscopic remission does not guarantee subsequent remission. Up to 50% of individuals with endoscopic remission on their index postoperative colonoscopy can experience disease progression regardless of medical therapy utilization with median time to progression of 18 months.18 Thus, those without endoscopic recurrence could be monitored with serial FCP every 3-6 months, periodic IUS, and ongoing colonoscopy surveillance in 1-2 years with subsequent intervals determined by findings.
Symptoms that mimic active Crohn’s disease can occur following an ileocecal resection and it is important for providers to understand possible etiologies and diagnostic plans. Postsurgical abdominal pain or discomfort is common in the days to weeks following the event, but typically steadily dissipates with time. Non-Crohn’s potential pain etiologies to be considered include postoperative complications (anastomotic leak, abscess, hematoma), impaired gastrointestinal motility (e.g. ileus, opioid-induced constipation or gastroparesis), adhesive disease, cholelithiasis, cholecystitis, nephrolithiasis, or urinary tract infections. Increased frequency and loose consistency of bowel movements can be normal gastrointestinal consequences of a resection surgery and intestinal adaption can occur in the months following. Fiber supplementation can often improve this clinical situation. Other postoperative diarrheal states should also be considered including Clostridium difficile infection, bile acid diarrhea secondary to ileal resection, and small intestinal bacterial overgrowth. Stool pathogen panels, glucose or lactulose breath testing, and a trial of bile acid sequestrant (e.g., cholestyramine, colestipol) can be considered. Finally, individuals with extensive or multiple prior resections may be at risk of short gut syndrome with consequent malabsorptive diarrhea, dehydration, weight loss, and electrolyte disturbances.
Postoperative health maintenance should include periodic assessments of nutritional status including Vitamin B12 and Vitamin D, immunization considerations for those on advanced therapy, monitoring weight and dietary intake, smoking cessation when applicable, and ensuring execution of the postoperative Crohn’s disease management and monitoring plan.
Conclusions
Despite medical and management advances, a significant portion of CD patients require resective surgery. Postoperative recurrence of CD is common, often silent, and requires appropriate therapeutic and monitoring strategies to prevent disease progression. Preoperative risk stratification can help identify patients who may benefit most from prophylactic medical therapy postoperatively. To date, anti-TNFs remain the most effective and studied therapy for prevention of Crohn’s disease in high-risk patients, however, vedolizumab is emerging as an option. Ongoing surveillance with fecal calprotectin and IUS, when available, and colonoscopy at 6-12 months postoperatively allows for early recurrence identification and treatment. There remain many key knowledge gaps in risk factors, biomarkers, and management algorithms for postoperative Crohn’s disease.
Financial Disclosures
Dr. Click – Consulting for AbbVie
Dr. Regueiro – Advisory Boards and Consultant (both) for Abbvie, Johnson and Johnson, UCB, Takeda, Pfizer, BMS, Organon, Amgen, Genentech, Gilead, Salix, Prometheus, Lilly, Celgene, Boehringer Ingelheim Pharmaceuticals Inc. (BIPI), Celltrion, Roche, Merck, Sanofi, Biocon, Abavax
References
References
1. Ananthakrishnan AN, Adler J, Chachu KA, et al. AGA Clinical Practice Guideline on the Role of Biomarkers for the Management of Crohn’s Disease. Gastroenterology. Dec 2023;165(6):1367–1399. doi:10.1053/j.gastro.2023.09.029
2. Bohra A, Van Langenberg DR, Vasudevan A. Intestinal Ultrasound in the Assessment of Luminal Crohn’s Disease. Gastrointestinal Disorders. 2022;4(4):249–262.
3. De Cruz P, Kamm MA, Prideaux L, Allen PB, Desmond PV. Postoperative recurrent luminal Crohn’s disease: a systematic review. Inflamm Bowel Dis. Apr 2012;18(4):758-77. doi:10.1002/ibd.21825
4. Shah RS, Nakamura T, Bachour S, et al. S0825 rior Surgical History Is the Strongest Risk Factor for Postoperative Crohn’s Disease Recurrence: A Guideline-Based Risk-Stratified Analysis. Official journal of the American College of Gastroenterology | ACG. 2020;115:S424. doi:10.14309/01.ajg.0000705348.73867.ec
5. Simillis C, Jacovides M, Reese GE, Yamamoto T, Tekkis PP. Meta-analysis of the role of granulomas in the recurrence of Crohn disease. Dis Colon Rectum. Feb 2010;53(2):177-85. doi:10.1007/DCR.0b013e3181b7bfb0
6. Li Y, Stocchi L, Rui Y, Remzi FH, Shen B. Comparable outcomes of the consistent use versus switched use of anti- tumor necrosis factor agents in postoperative recurrent Crohn’s disease following ileocolonic resection. Int J Colorectal Dis. Nov 2016;31(11):1751-1758. doi:10.1007/s00384-016-2632-4
7. Singh S, Garg SK, Pardi DS, Wang Z, Murad MH, Loftus EV, Jr. Comparative efficacy of pharmacologic interventions in preventing relapse of Crohn’s disease after surgery: a systematic review and network meta-analysis. Gastroenterology. Jan 2015;148(1):64–76 e2; quiz e14. doi:10.1053/j.gastro.2014.09.031
8. Regueiro M, Feagan BG, Zou B, et al. Infliximab Reduces Endoscopic, but Not Clinical, Recurrence of Crohn’s Disease After Ileocolonic Resection. Gastroenterology. Jun 2016;150(7):1568-1578. doi:10.1053/j.gastro.2016.02.072
9. Papamichael K, Archavlis E, Lariou C, Mantzaris GJ. Adalimumab for the prevention and/or treatment of post-operative recurrence of Crohn’s disease: a prospective, two-year, single center, pilot study. J Crohns Colitis. Oct 2012;6(9):924-31. doi:10.1016/j.crohns.2012.02.012
10. Savarino E, Bodini G, Dulbecco P, et al. Adalimumab is more effective than azathioprine and mesalamine at preventing postoperative recurrence of Crohn’s disease: a randomized controlled trial. Am J Gastroenterol. Nov 2013;108(11):1731-42. doi:10.1038/ajg.2013.287
11. Savarino E, Dulbecco P, Bodini G, Assandri L, Savarino V. Prevention of postoperative recurrence of Crohn’s disease by Adalimumab: a case series. Eur J Gastroenterol Hepatol. Apr 2012;24(4):468-70. doi:10.1097/MEG.0b013e3283500849
12. D’Haens G, Taxonera C, Lopez-Sanroman A, et al. Vedolizumab to prevent postoperative recurrence of Crohn’s disease (REPREVIO): a multicentre, double-blind, randomised, placebo-controlled trial. Lancet Gastroenterol Hepatol. Jan 2025;10(1):26-33. doi:10.1016/s2468-1253(24)00317-0
13. De Cruz P, Kamm MA, Hamilton AL, et al. Crohn’s disease management after intestinal resection: a randomised trial. Lancet. Apr 11 2015;385(9976):1406-17. doi:10.1016/s0140-6736(14)61908-5
14. Joustra V, van Sabben J, van der Does de Willebois E, et al. Benefit of Risk-stratified Prophylactic Treatment on Clinical Outcome in Postoperative Crohn’s Disease. J Crohns Colitis. Apr 3 2023;17(3):318–328. doi:10.1093/ecco-jcc/jjac139
15. Ten Bokkel Huinink S, Bak MTJ, Beelen EMJ, et al. The Impact of Postoperative Prophylactic Medication on Long-Term Surgical, Severe Endoscopic and Endoscopic or Radiologic Recurrence Following Primary Ileocecal Resection in Patients With Crohn’s Disease. Aliment Pharmacol Ther. Mar 2025;61(6):1019–1031. doi:10.1111/apt.18496
16. Arkenbosch JHC, Beelen EMJ, Dijkstra G, et al. Prophylactic Medication for the Prevention of Endoscopic Recurrence in Crohn’s Disease: a Prospective Study Based on Clinical Risk Stratification. J Crohns Colitis. Mar 18 2023;17(2):221–230. doi:10.1093/ecco-jcc/jjac128
17. Cohen BL, Fleshner P, Kane SV, et al. Anti-Tumor Necrosis Factor Therapy is Not Associated with Post-Operative Infection: Results from Prospective Cohort of Ulcerative Colitis and Crohn’s Disease Patients Undergoing Surgery to Identify Risk Factors for Postoperative Infection I (Puccini). Gastroenterology. 2019;156(6):S-80. doi:10.1016/S0016-5085(19)36987-2
18. Pouillon L, Remen T, Amicone C, et al. Risk of Late Postoperative Recurrence of Crohn’s Disease in Patients in Endoscopic Remission After Ileocecal Resection, Over 10 Years at Multiple Centers. Clin Gastroenterol Hepatol. Jun 2021;19(6):1218-1225.e4. doi:10.1016/j.cgh.2020.05.027
 Benjamin Click
Benjamin Click Miguel Regueiro
Miguel Regueiro Alexander W. Worix
Alexander W. Worix Edwin McDonald
Edwin McDonald Taranika Sarkar Das
Taranika Sarkar Das Douglas G. Adler
Douglas G. Adler
 Tasnim Ahmed
Tasnim Ahmed Stephen Le Breton
Stephen Le Breton Tyler M. Berzin
Tyler M. Berzin

 Uma Mahadevan
Uma Mahadevan


 Christina M. Minami
Christina M. Minami


 Neha D. Shah
Neha D. Shah Elizabeth Wall
Elizabeth Wall