Effective management of Disorders of Gut-Brain Interaction (DGBIs) often requires an interdisciplinary approach that extends beyond the gastrointestinal (GI) provider. DGBIs are characterized by dysregulation of the gut–brain axis, and growing evidence supports behavioral and dietary therapies that target these bidirectional pathways. Behavioral health providers and registered dietitians (RD) with specialized gastroenterology training — hereafter referred to as GI-psychologists and GI-RDs — deliver evidence-based interventions that have potential to improve patient symptoms and quality of life (QoL), with GI-psychologists using Brain-Gut Behavioral Therapies (BGBT), such as cognitive-behavioral therapy, to address psychosocial contributors, and GI-RDs applying diet strategies, including the low FODMAP diet, to optimize gastrointestinal function. Collaboration between these providers enables comprehensive evaluation of symptoms and DGBI subtype, supporting appropriate referrals. This review is to summarize current evidence for therapy approaches and delineates the distinct and complementary contributions of GI-psychologists and GI-RDs in DGBI, with the aim of guiding interdisciplinary referral practices.
The Role of the Brain-Gut Axis in Disorders of Gut-Brain Interactions
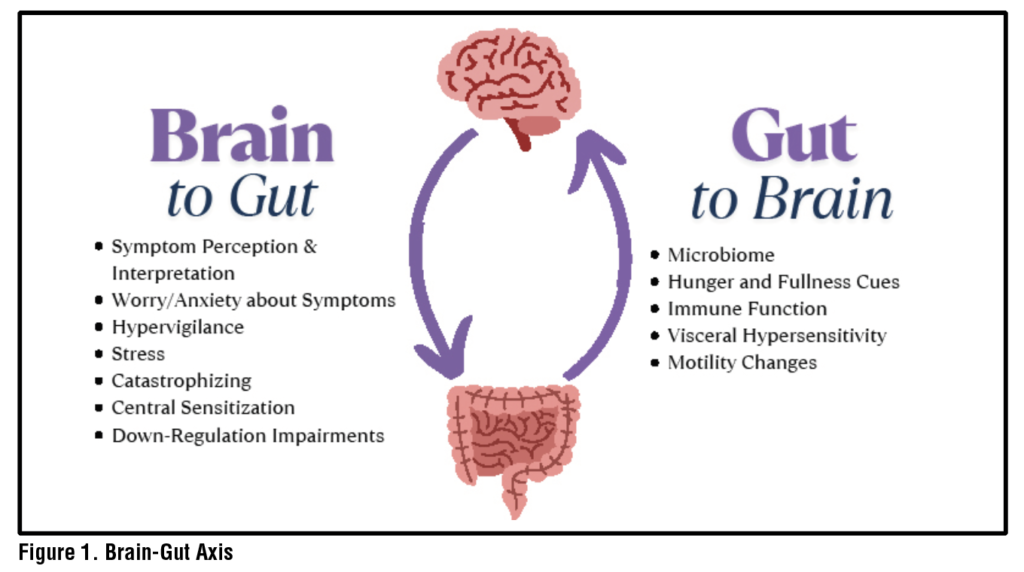
Symptoms and sensations in the digestive tract can be related to structural organ malfunction or from sensory malfunction via the Gut-Brain Axis (GBA). The central nervous and enteric nervous systems are connected to one another via complex nervous, endocrine and immune bidirectional pathways.1–3. When the GBA is dysregulated, benign sensations in the GI organs such as those of normal digestive processes may be perceived as threatening and more severe due to cognitive, behavioral and affective processes which can function to amplify those signals.4 For example, relevant processes often include symptom or illness specific anxiety and catastrophizing, avoidance of feared activities and foods, hypervigilance to bodily sensations, and activation of the stress response.5,6 These factors interact with central processes which contribute to symptoms including visceral hypersensitivity of the peripheral nerves in the digestive tract and to enhanced pain perception through central sensitization.7 (See Figure 1) Disorders derived from disruption of the GBA formerly called Functional Gastrointestinal (GI) Disorders and are now termed Disorders of Gut-Brain Interaction (DGBI) (e.g., irritable bowel syndrome (IBS), functional dyspepsia).8
A challenge for many patients with DGBI driven symptoms is a lack of a clear mechanism for their concern, as typically objective testing does not provide an explanation. An interdisciplinary GI team can engage in a collaborative discussion starting from a clear explanation of the GBA, what DGBI diagnosis they meet criteria for, and what evidence-based treatments they may qualify for to manage and treat their symptoms with the various specialists available to them.9 This can offer much needed assurance that the symptoms are real and expected in DGBI, can help reduce stigma associated with these disorders and can help patients develop a clear treatment plan.10
Brain-Gut Behavioral Therapies
When accessible and appropriate for the patient, Brain-Gut Behavioral Therapies (BGBTs) can offer a pathway to symptom relief for patients with DGBIs. To date, BGBTs with a robust evidence base include Cognitive Behavioral Therapy (CBT), gut-directed hypnotherapy (GDH)11 and mindfulness therapies such as Acceptance and Commitment Therapy (ACT).12 (See Table 1) The aim of these therapies is to target the functioning of the brain and gut through modifying underlying interpretations about symptoms and illness.
Cognitive Behavioral Therapy
CBT is a skills-based therapy which emphasizes the interconnectedness of thoughts, emotions, behaviors, and physical sensations. While CBT was initially developed and studied in mental health, it has been successfully applied in medical settings including DGBI. Components of the treatment vary based on patient presentation but often include relaxation strategies (including instruction in diaphragmatic breathing), cognitive awareness and reframing, exposure exercises to avoided foods or situations and stress management with problem solving skills. Specifically in IBS, CBT has demonstrated benefit to symptom experience and severity,quality of life and impact on the brain’s interpretation of symptoms.13–17 For non-cardiac chest pain and functional dyspepsia, smaller studies have also shown benefit in symptom perception.18,19 CBT has also been recommended as an intervention target for functional heartburn, though robust trials to test efficacy are still needed.20 Beyond traditional face to face delivery, evidence suggests that CBT for IBS can be successfully administered in group format and online/telephone.21
Table 1. Evidence-based Treatments: Cognitive Behavioral Therapy, Gut-Directed Hypnotherapy, and Acceptance and Commitment Therapy
| Evidence Based Treatment for DGBI | Main Components | Typical Course | Patient Characteristic Considerations |
| Cognitive Behavioral Therapy (CBT)15,21,46 | An intervention which includes psychoeducation about the condition and gut-brain axis, skills training to target and change maladaptive cognitions, skills training to target physiological arousal and promote relaxation, and strategies to help broaden behavioral responses to symptoms and interoceptive and behavioral exposure. | ~3-12 sessions | Patient endorses symptoms specific anxiety and behavioral avoidance. Patient is psychologically minded, meaning they are interested and able to see themselves and their own thoughts and behavior from a 3rd person perspective, analyzing these and apply skills to make change. |
| Gut-Directed Hypnotherapy23,27,47 (GDH) | A facilitated state of deep relaxation, known as trance, in which patients have increased receptiveness to suggestion. Suggestions are tailored to patient’s specific symptoms and quality of life concerns. | ~4-12 sessions | Patient endorses pain, tension/tightness, or visceral hypersensitivity as a primary symptom/experience. Patients do NOT have active symptoms of post-traumatic stress disorder (PTSD), or PTSD symptoms are stable at present. |
| Acceptance and Commitment Therapy (ACT)30,48 | Identification of valued life domains and explicit formulation of committed actions patient can take to live life in greater accordance with their values, mindfulness practice, reduction of emotional avoidance, identification of “fusion” with maladaptive thoughts and skills taught to help “de-fuse” from these thoughts. | ~5-12 sessions | Patient endorses difficulty putting space between themselves and negative thoughts and emotions related to GI symptoms or condition or is feeling “stuck.” Patient is noting that their GI condition or symptoms are preventing them from living life in accordance with the values that are important to them. |
Gut-Directed Hypnotherapy
GDH is a form of therapy that, over a series of sessions, guides patients into a deep relaxation and further into a hypnotic or trance state prior to the delivery of tailored suggestions to modify their visceral sensations and pain experience. Therapeutic suggestions also often include those to increase patient’s engagement in valued life activities over attending to bodily sensations. Frequent practice, often with audio, is a common component of the therapy. GDH has shown to have a substantial impact on IBS symptoms and abdominal pain.22,23 Smaller studies examining GDH in the esophagus, such as in functional heartburn and globus, have also shown promise for improving symptoms but warrant replication in larger trials.24–26 Like CBT, GDH can be effectively delivered in a variety of modalities including via video and in groups.23,27,28
Table 2. Nutrition Care Plan: The GI-RDs Contribution to the Interdisciplinary Team41-43
| Nutrition Assessment Components | RD Considerations | Nutrition Interventions/GI-RD “toolbox” | Nutrition Monitoring and Evaluation |
| Nutritional Status | Weight BMI Weight History Malnutrition Appropriate labs (e.g., prealbumin, electrolytes, zinc, iron studies) Nutrition Focused Physical Exam (NFPE) | Weight restoration with slow caloric increase; may use oral nutrition supplement as first line treatment Address micronutrient concerns with supplementation Nutrition education focused on role of weight restoration on GI symptom management | Weight gain Dietary components (e.g., % of estimated energy needs in dietary intake, fiber intake, micronutrient intake) Labs Improvement of muscle wasting & fat loss on NFPE |
| Nutrition/Food History | Previous diet therapy attempts (Gluten-Free Diet (GFD), Low FODMAP Diet, National Institute for Health and Care Excellence (NICE), etc.) Herbal supplement usage History of eating disorder (ED) for shape/weight concerns Active ED Current diet restrictions are significant enough to cause concern for diet quality Prescence and severity of sitophobia Assess for avoidant restrictive food intake disorder (ARFID). Consider using ARFID screener, though not currently validated in the GI patient population specifically (e.g., PARDI-AR-Q,49 Nine Item ARFID Screen43) | Pausing other supplements; trial of peppermint oil,50 melatonin,51 psyllium husk52 Personalized therapeutic dietary interventions that could include changes in eating behaviors and meal timing, consistent fiber intake, FODMAP restrictions among others For patients with a history of ED, consider gentle nutrition, modified FODMAP diet, as well as mindful eating Triage level of care to eating disorder treatment Referral to GI-psychologist for additional food exposure support for those with sitophobia Develop food hierarchy and exposures trials in tandem with GI-psychologist in mild sitophobia or ARFID Refer to an ARFID treatment center for patients with moderate-severe ARFID, AFTER evaluation by GI-psychologist Nutrition counseling: Optimize nutritional intake and health, improve or stabilize GI symptoms as part of DGBI while providing validation | GI symptom frequency GI symptom severity Diet quality Development of maladaptive dietary behaviors e.g., over restriction, skipping meals, reducing intake to <75% of estimated energy needs |
| Psychosocial History | Mental health Resources, including financial, time, logistical Motivation Social factors, environment Cultural factors Health literacy | Collaboration of care meeting with current therapist, and/or refer to general mental health community provider Motivational interviewing Nutrition education materials appropriate for knowledge level Meal delivery services, brands, recipes developed appropriate for diet application if kitchen skills are limited | Readiness to change Knowledge recall |
Acceptance and Commitment Therapy
Mindfulness based therapies, such as ACT, aim to assist patients in finding grounding in the present moment in order to experience the transient nature of thoughts, emotions and urges, a process known as psychological flexibility.29 In DGBI, this may look like a patient acknowledging that their symptoms are present today, accepting that it is frustrating and ultimately choosing to attend an important social gathering and being open to finding joy in that experience. Another key component of ACT is assisting patients in identifying their personal values and finding opportunities to commit to behaviors and actions that are consistent with those values. In general, the literature on ACT as a BGBT for DGBI is more nascent than that of CBT or GDH and deserves further study. However, those studies implementing a full ACT protocol for patients with DGBI, namely, IBS, have found ACT may help reduce symptom severity and acceptance of their diagnosis.30
Though all the therapies are described separately above, it is not uncommon for a skilled provider to use one or multiple BGBT skills during the intervention course with a patient.
Diet Therapy in DGBI
When patients are asked about their preference of medical, dietary or behavioral intervention, one study found that patients with DGBI prefer diet-focused interventions as first line therapy.31 The American Gastroenterological Association 2022 Clinical Practice Update on the Role of Diet in Irritable Bowel Syndrome highlighted several best practice advice statements that focus on diet and the role of a dietitian in IBS care.32 These include nutrition assessment and screening for eating disorders prior to dietary restriction, providing nutrition education about the role of food and meal-related symptoms, and personalization of meal choices. Instructing patients to keep a 3-day food and symptom log prior to the first GI-RD visit may help illuminate these patterns.32
Therapeutic diets, such as low FODMAP diet that have shown efficacy in reducing symptoms of IBS, should be used for a finite period and may not be an appropriate starting point for patients who are consuming low culprit foods, have active eating, or psychiatric disorders or are food insecure.32 Additionally, alterations of the microbes of the gut, food chemistry and GI infections have been identified as mechanisms which have potential to increase intestinal permeability and hypersensitivity in DGBI.3,33,34 Recent understanding of gut microbiome and its role in the GBA suggest that therapeutic diets may have several impacts by directly modulating both the microbiota and its metabolome which play a role in the GBA.35
Table 3. DGBI Counseling Services Referral Considerations
| DGBI Patient Presentation | Psychology Referral | Dietitian Referral |
| Observed association between higher levels of stress and worsening GI symptoms, regardless of diet consumption | X | |
| Presence of or anticipation of GI symptoms creates worry or anxiety | X | |
| Significantly changed behavior in an effort to control symptoms | X | X |
| Avoidance of eating foods in general or broad general classes of foods | X | X |
| Discordance between objective, diagnostic testing and patients report of symptoms | X | |
| Meal-related GI symptoms | X | |
| Specific concern for dietary Intolerance, such as carbohydrate malabsorption or gluten intolerance | X | |
| Unintentional weight loss and/or a concern for malnutrition | X | X |
| History of disordered eating in newly diagnosed DGBI | X | X |
| Nutritionally pertinent medical diagnosis, such as diabetes, CKD, CVD | X | |
| Interested in holistic lifestyle-based therapies vs. medication, or in addition to medical therapy | X | X |
Role of GI-Psychologist: Assessment and Treatment Options
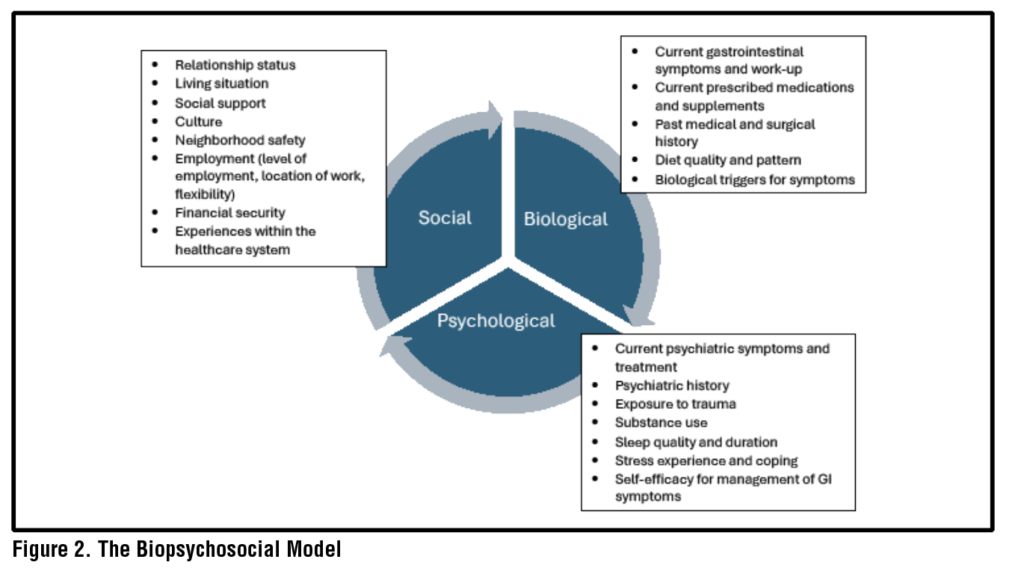
GI-psychologists, with clinical skills and specialized training in the functioning of the GI tract, are well positioned to apply BGBTs. The process begins with an evaluation to ascertain components of the biopsychosocial model.36–38 (See Figure 2) Patients are typically asked to provide a timeline of their GI symptom onset, as well as potential gut-brain dysregulation triggers, such as stressful life events, exposure to physical (e.g., abdominal surgery) or psychological (e.g., sexual abuse) trauma, or the experience of infections. The GI-psychologist seeks to understand progression of symptoms over time, including whether symptoms worsen with higher levels of stress, and how GI symptoms or their management impact QoL, functioning, and relationships, with attention to symptom-specific anxiety, hypervigilance, and visceral hypersensitivity.25
During the evaluation, the GI-psychologist gathers information on life domains that may affect GI symptoms. Patients are typically asked about any current and past psychiatric symptoms and history, including frank and subclinical eating disorders, psychotherapy, and psychotropic medications.39 Accordingly, patients are also asked to describe their global levels of perceived stress and the coping strategies they may use to handle stress to help determine an approach versus avoidance style. Physical health behaviors, including use of substances like alcohol, cannabis and nicotine, quality and duration of sleep, typical diet, food triggers, and engagement of physical activity are reviewed for potential to worsen symptoms. Patients assigned female at birth may also be asked questions in the evaluation regarding reproductive health, such as whether GI symptoms coincide or worsen with menstrual cycles. Typically, the assessment will include conceptualization, treatment planning, and potential referrals to outside providers.
The GI-psychologist may refer patients to external providers and services needed to augment their care. Among others, these commonly include community resources, pelvic-floor physical therapy (PFPT), specialists or programs for eating disorders (e.g., laxative abuse, severe restriction with or without body and shape concerns), or specialists in psychiatric concerns. Depending on the symptom severity of a patient’s mental health presentation, the GI-psychologist will collaborate with the patient to determine the sequence of treatment, i.e., whether BGBT can occur in parallel to or after a patient’s primary mental health or eating disorder treatment.
Similarly, the GI-psychologist may make a referral to a GI-RD to help elucidate various components of diet, nutritional status and if a therapeutic diet trial is warranted.40
Role of GI-RD: Assessment and Treatment Options
The first step with a GI-RD is the initial visit, during which the dietitian conducts a nutrition assessment to obtain detailed information regarding a patient’s medical history, nutrition history, diet recall, lifestyle habits, cultural considerations and knowledge, beliefs, and attitudes regarding food. Specifically, a GI-RD closely evaluates presentation of a patient’s symptoms, appropriateness of current diet, past or current eating behaviors that may indicate a disordered relationship with food, known food allergies, intolerances, or sensitivities, and the extent of dietary restriction.
Based on these findings, the GI-RD collaborates on a treatment plan and provides tailored recommendations for nutrition interventions to treat the DGBI. Follow-up visits with a GI-RD include assessments of the outcome of the initial nutrition interventions, adjusts as needed, and provide ongoing education and counseling to optimize progress. If the GI-RD identifies factors that inform the need for a referral to a GI-psychologist, the dietitian may make a direct referral to support integrative care.41–43 (See Table 2)
Intersection and Overlap of GI-Psychologist and GI-RD
In an interdisciplinary care model, GI-RDs’ and GI-psychologists’ roles often overlap. It can be challenging to define the boundaries of nutrition interventions centered on diet as providers of both disciplines have applicable skillsets. A collaborative treatment plan and active communication regarding a patient’s goals and intervention barriers and facilitators can be exceptionally helpful in these situations to enhance progress. Importantly, medical therapy is less than 50% effective at treating global GI symptoms, suggesting the necessity of several modalities to best address both GI and extra-intestinal symptoms.44
Food and eating play a critical role in the experience of GI symptoms; beliefs about food, eating and digestion can trigger maladaptive processes such as symptom-specific anxiety, avoidance of food(s), and hypervigilance to the body after eating, reinforcing the DGBI. Approximately 80% of patients with DGBI implicate food as a catalyst for symptom onsetand develop adaptive eating behaviors to manage symptoms.40,45 Inappropriate avoidance of foods can lead to overly restricted diets and malnutrition. For example, patients with sitophobia often have highly restricted diets and struggle to reintroduce a wider variety of foods, even after food intolerances and allergies have been ruled out. A GI-psychologist and GI-RD team can collaborate to create a food avoidance hierarchy and assist the patient in utilizing psychological and behavioral skills to incorporate foods systematically. Other considerations for referrals are outlined in Table 3.
Case Scenario and Treatment Course
To further illustrate interdisciplinary care and the components of treatment, please see a case example:
Case:
Patient is a 45-year-old white, married straight cis-woman with IBS-C, gastritis with possible GERD (awaiting objective testing). She is 20 years post-cholecystectomy.
The patient is currently experiencing days of constipation followed by a period of diarrhea, post-prandial abdominal discomfort, nausea, heartburn and bloating. She is taking plecanatide (3mg) and omeprazole (20mg). Prior therapeutics include linaclotide, lubiprostone, prucalopride, cholestyramine, and nortriptyline.
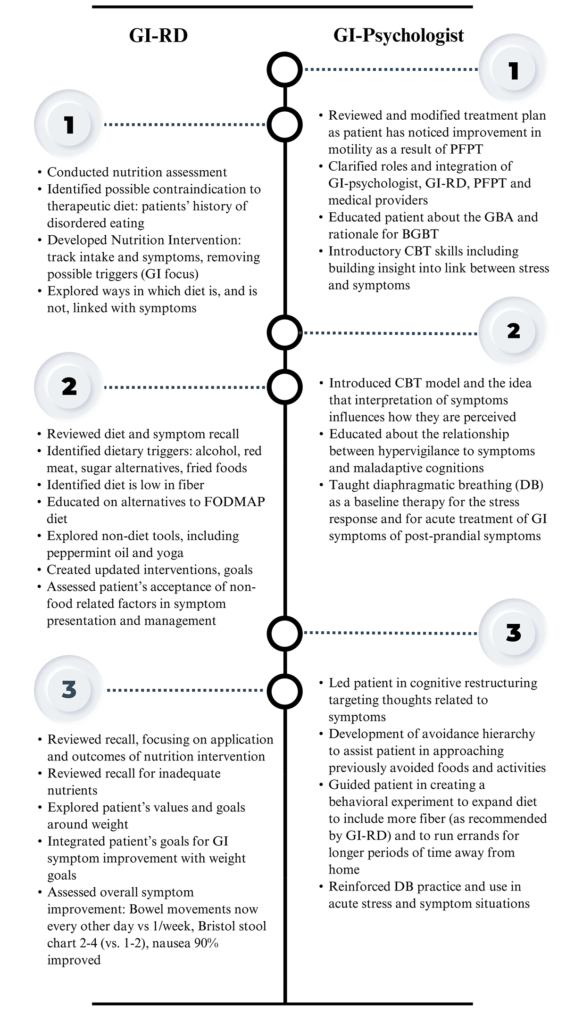
Table 4. Case Scenario. Between GI-Psych Evaluation and intervention session #1, the patient had consultation with a motility specialist, underwent anorectal manometry (ARM), was referred and started pelvic floor physical therapy (PFPT) and scheduled consultation with GI-RD.
Patient was referred to GI-psychologist by her general gastroenterologist for treatment of IBS-C and other GI symptoms. Condensed and pertinent data from that evaluation is below:
Patient reported that lower GI symptoms began over 20 years ago, around the time she was in an abusive relationship. In the last several years, her upper GI and abdominal symptoms have worsened. In addition to symptoms, she reports a concern about obtaining an accurate diagnosis that captures the range of problems with her GI system. She described several factors that are likely contributing to current symptoms and associated distress including long history of anxiety, OCD and experience of trauma which may contribute to GBA disruption.
The patient described vigilance to her body sensations, her body weight/shape and to her diet as her symptoms occur reliably after eating. This is further distressing to her as she has noticed weight gain in recent years. The patient described a tendency to avoid leaving home for fear of not knowing where bathrooms will be. She described a strong relationship between stress and GI symptoms and noted an overall high level of stress which she attributed to her own anxiety and tendency toward perfectionism and people-pleasing. She reported no concerns regarding social influences on health.
Patient reported treatment goals are to:
- Understand etiology of symptoms
- Improve consistency of her bowel movements
- Understand dietary triggers
- Decrease worries and anxiety about her symptoms and increase comfort leaving home
- Reduce weight and bloating as this is a body image concern
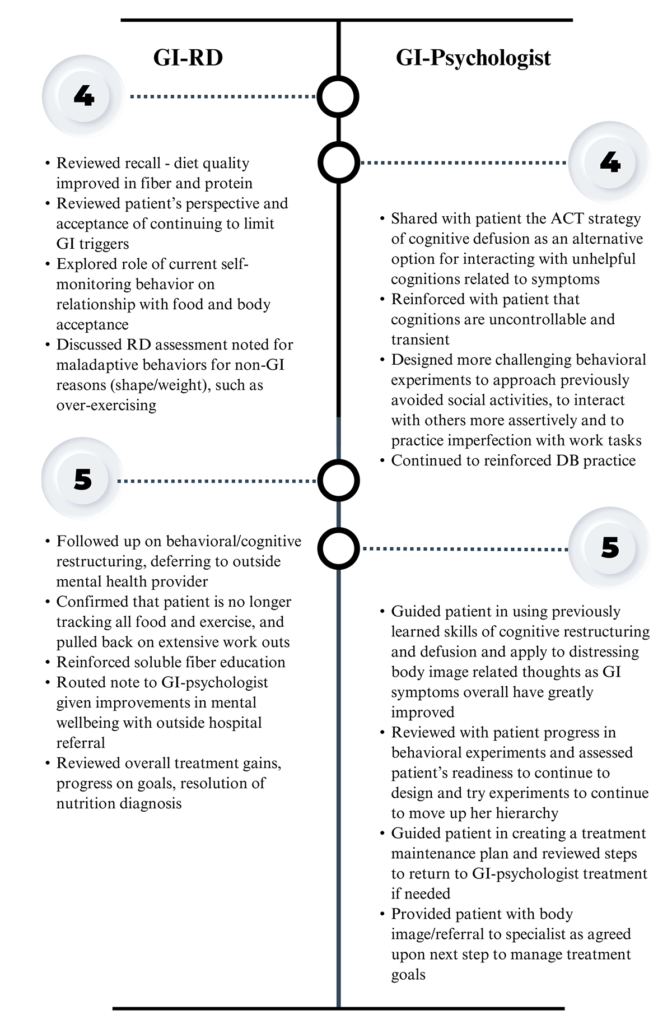
Table 4. Case Scenario (Continued ). Following her engagement with a multi-disciplinary treatment team including motility specialist, PFPT, GI-psychologist and GI-RD, patient reported a significant improvement in symptoms of constipation and only occasional post-prandial abdominal discomfort, nausea, heartburn and bloating. She expressed satisfaction with response to treatment and the integration of her care team. At this point, GI-psychologist and GI-RD terminated care as treatment goals had been met.
GI-psychologist next steps:
- Request referral to motility specialist to evaluate for pelvic floor disorder given patient’s experience of alternating constipation and diarrhea
- Referral to GI-RD as patient is experiencing post-prandial symptoms
- Referral to a trauma specialist as a patient is experiencing current post-traumatic stress symptoms
- Plan for treatment including CBT and ACT. Plan to defer gut-directed hypnotherapy as patient is experiencing current trauma symptoms
See Table 4 for the patient’s treatment course with GI-psychologist and GI-RD working as a team in integrated care.
References
1. Agirman G, Yu KB, Hsiao EY. Signaling inflammation across the gut-brain axis. Science. 2021;374(6571):1087-1092.
2. Palsson OS, Ballou S. Hypnosis and cognitive behavioral therapies for the management of gastrointestinal disorders. Curr Gastroenterol Rep. 2020;22(7):1-9.
3. Mayer EA, Nance K, Chen S. The gut-brain axis. Annu Rev Med. 2022;73:439-453.
4. Labanski A, Langhorst J, Engler H, Elsenbruch S. Stress and the brain-gut axis in functional and chronic-inflammatory gastrointestinal diseases: A transdisciplinary challenge. Psychoneuroendocrinology. 2020;111:104501.
5. Labus JS, Bolus R, Chang L, et al. The Visceral Sensitivity Index: development and validation of a gastrointestinal symptom-specific anxiety scale. Aliment Pharmacol Ther. 2004;20(1):89-97.
6. Ballou S, Vasant DH, Guadagnoli L, et al. A primer for the gastroenterology provider on psychosocial assessment of patients with disorders of gut-brain interaction. Neurogastroenterol Motil. 2024;36(12):e14894.
7. Keefer L, Ko CW, Ford AC. AGA clinical practice update on management of chronic gastrointestinal pain in disorders of gut-brain interaction: Expert review. Clin Gastroenterol Hepatol. 2021;19(12):2481-2488.e1.
8. Schmulson MJ, Drossman DA. What is new in Rome IV. Journal of Neurogastroenterology and Motility. 2017;23(2):151-163.
9. Keszthelyi D, Keefer L. “A rose by any other name”? — Beyond diagnostic nomenclature in neurogastroenterology. Gastroenterology. 2025;169(1):19-21. doi:10.1053/j.gastro.2025.01.229
10. Feingold JH, Drossman DA. Deconstructing stigma as a barrier to treating DGBI: Lessons for clinicians. Neurogastroenterol Motil. 2021;33(2):e14080.
11. Black CJ, Thakur ER, Houghton LA, Quigley EMM, Moayyedi P, Ford AC. Efficacy of psychological therapies for irritable bowel syndrome: systematic review and network meta-analysis. Gut. 2020;69(8):1441-1451.
12. Keefer L, Ballou SK, Drossman DA, Ringstrom G, Elsenbruch S, Ljótsson B. A Rome working team report on brain-gut behavior therapies for disorders of gut-brain interaction. Gastroenterology. 2022;162(1):300-315.
13. Goodoory VC, Khasawneh M, Thakur ER, et al. Effect of brain-gut behavioral treatments on abdominal pain in irritable bowel syndrome: Systematic review and network meta-analysis. Gastroenterology. 2024;167(5):934-943.e5.
14. Bonnert M, Olén O, Lalouni M, et al. Internet-delivered cognitive behavior therapy for adolescents with irritable bowel syndrome: A randomized controlled trial. Official journal of the American College of Gastroenterology | ACG. 2017;112(1):152.
15. Owusu JT, Sibelli A, Moss-Morris R, van Tilburg MAL, Levy RL, Oser M. A pilot feasibility study of an unguided, internet-delivered cognitive behavioral therapy program for irritable bowel syndrome. Neurogastroenterol Motil. 2021;33(11):e14108.
16. Hunt M, Miguez S, Dukas B, Onwude O, White S. Efficacy of Zemedy, a mobile digital therapeutic for the self-management of irritable bowel syndrome: crossover randomized controlled trial. JMIR Mhealth Uhealth. 2021;9(5):e26152.
17. Jacobs JP, Gupta A, Bhatt RR, et al. Cognitive behavioral therapy for irritable bowel syndrome induces bidirectional alterations in the brain-gut-microbiome axis associated with gastrointestinal symptom improvement. Microbiome. 2021;9(1):236.
18. Jonsbu E, Martinsen EW, Morken G, Moum T, Dammen T. Change and impact of illness perceptions among patients with non-cardiac chest pain or benign palpitations following three sessions of CBT. Behavioural and Cognitive Psychotherapy. 2013;41(4):398-407.
19. Keefer L, Ballou SK, Drossman DA, Ringstrom G, Elsenbruch S, Ljótsson B. A Rome working team report on brain-gut behavior therapies for disorders of gut-brain interaction. Gastroenterology. 2022;162(1):300-315.
20. Guadagnoli L, Yadlapati R, Pandolfino J, et al. Behavioral therapy for functional heartburn: Recommendation statements. Clin Gastroenterol Hepatol. 2024;22(8):1709-1718.e3.
21. Chen LJ, Kamp K, Fang A, Heitkemper MM. Delivery methods of cognitive behavior therapy for patients with irritable bowel syndrome. Gastroenterol Nurs. 2022;45(3):149-158.
22. Lövdahl J, Törnblom H, Ringström G, Palsson OS, Simrén M. Randomised clinical trial: individual versus group hypnotherapy for irritable bowel syndrome. Aliment Pharmacol Ther. 2022;55(12):1501-1511.
23. Adler EC, Levine EH, Ibarra AN, et al. Gut-directed hypnotherapy for irritable bowel syndrome: A systematic review and meta-analysis. Neurogastroenterology & Motility. 2025;37(7):e70037.
24. Kiebles JL, Kwiatek MA, Pandolfino JE, Kahrilas PJ, Keefer L. Do patients with globus sensation respond to hypnotically assisted relaxation therapy? A case series report. Accessed January 30, 2026. https://dx.doi.org/10.1111/j.1442-2050.2010.01064.x
25. Riehl ME, Kinsinger S, Kahrilas P, Pandolfino J, Keefer L. The role of a health psychologist in the management of functional esophageal complaints. Dis Esophagus. 2015;28(5):428-436.
26. Riehl ME, Pandolfino JE, Palsson OS, Keefer L. The feasibility and acceptability of esophageal-directed hypnotherapy for functional heartburn. Dis Esophagus. 2016;29(5):490-496.
27. Anderson EJ, Peters SL, Gibson PR, Halmos EP. Comparison of digitally delivered gut-directed hypnotherapy program with an active control for irritable bowel syndrome. Official journal of the American College of Gastroenterology | ACG. 2025;120(2):440.
28. Peters SL, Gibson PR, Halmos EP. Smartphone app-delivered gut-directed hypnotherapy improves symptoms of self-reported irritable bowel syndrome: A retrospective evaluation. Neurogastroenterol Motil. 2023;35(4):e14533.
29. Hayes SC, Levin ME, Plumb-Vilardaga J, Villatte JL, Pistorello J. Acceptance and commitment therapy and contextual behavioral science: Examining the progress of a distinctive model of behavioral and cognitive therapy. Behav Ther. 2013;44(2):180-198.
30. Marchese SH, Naftaly JP, Pandolfino J. Acceptance and commitment therapy for the treatment of irritable bowel syndrome and inflammatory bowel disease: a narrative review. Transl Gastroenterol Hepatol. 2024;9:43.
31. Sturkenboom R, Keszthelyi D, Masclee AAM, Essers BAB. Discrete choice experiment reveals strong preference for dietary treatment among patients with irritable bowel syndrome. Clinical Gastroenterology and Hepatology. 2022;20(11):2628-2637.
32. Chey WD, Hashash JG, Manning L, Chang L. AGA Clinical practice update on the role of diet in irritable bowel syndrome: Expert review. Gastroenterology. 2022;162(6):1737-1745.e5.
33. Barbara G, Aziz I, Ballou S, et al. Rome Foundation working team report on overlap in disorders of gut-brain interaction. Nat Rev Gastroenterol Hepatol. 2025;22(4):228-251.
34. Lacy BE, Pimentel M, Brenner DM, et al. ACG clinical guideline: Management of irritable bowel syndrome. Official journal of the American College of Gastroenterology | ACG. 2021;116(1):17.
35. Schneider E, O’Riordan KJ, Clarke G, Cryan JF. Feeding gut microbes to nourish the brain: unravelling the diet–microbiota–gut–brain axis. Nat Metab. 2024;6(8):1454-1478.
36. Engel GL. The need for a new medical model: A challenge for biomedicine. Science. 1977;196(4286):129-136.
37. Bolton D. A revitalized biopsychosocial model: core theory, research paradigms, and clinical implications. Psychological Medicine. 2023;53(16):7504-7511.
38. Van Oudenhove L, Levy RL, Crowell MD, et al. Biopsychosocial aspects of functional gastrointestinal disorders: How central and environmental processes contribute to the development and expression of functional gastrointestinal disorders. Gastroenterology. 2016;150(6):1355-1367.e2.
39. Kinsinger SW. Practical Approaches to Working with a gastrointestinal psychologist. Gastroenterol Clin North Am. 2022;51(4):711-721.
40. Böhn L, Störsrud S, Törnblom H, Bengtsson U, Simrén M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am J Gastroenterol. 2013;108(5):634-641.
41. Scarlata K, Catsos P, Smith J. From a dietitian’s perspective, diets for irritable bowel syndrome are not one size fits all. Clinical Gastroenterology and Hepatology. 2020;18(3):543-545.
42. Guadagnoli L, Mutlu EA, Doerfler B, Ibrahim A, Brenner D, Taft TH. Food-related quality of life in patients with inflammatory bowel disease and irritable bowel syndrome. Qual Life Res. 2019;28(8):2195-2205.
43. Murray HB, Dreier MJ, Zickgraf HF, et al. Validation of the Nine Item ARFID Screen (NIAS) subscales for distinguishing ARFID presentations and screening for ARFID. Int J Eat Disord. 2021;54(10):1782-1792.
44. Chey WD, Keefer L, Whelan K, Gibson PR. Behavioral and diet therapies in integrated care for patients with irritable bowel syndrome. Gastroenterology. 2021;160(1):47-62.
45. Scarlata K, Zickgraf HF, Satherley RM, et al. A call to action: Unraveling the nuance of adapted eating behaviors in individuals with gastrointestinal conditions. Clinical Gastroenterology and Hepatology. 2025;23(6):893-901.e2.
46. Sugaya N, Shirotsuki K, Nakao M. Cognitive behavioral treatment for irritable bowel syndrome: a recent literature review. BioPsychoSocial Med. 2021;15(1):1.
47. Sasegbon A, Hasan SS, Whorwell PJ, Vasant DH. Experience and clinical efficacy of gut-directed hypnotherapy in an Asian population with refractory irritable bowel syndrome. JGH Open. 2022;6(7):447-453.
48. Taghvaeinia A, Karami M, Azizi A. Comparison of the effect of dialectical behavior therapy, acceptance and commitment therapy mindfulness-based stress reduction on irritable bowel syndrome symptoms, quality of life, anxiety and depression: A pilot randomized controlled trial. Psychiatr Q. 2024;95(1):53-68.
49. Bryant-Waugh R, Stern CM, Dreier MJ, et al. Preliminary validation of the pica, ARFID and rumination disorder interview ARFID questionnaire (PARDI-AR-Q). J Eat Disord. 2022;10:179.
50. Alammar N, Wang L, Saberi B, et al. The impact of peppermint oil on the irritable bowel syndrome: a meta-analysis of the pooled clinical data. BMC Complement Altern Med. 2019;19:21.
51. Siah KTH, Wong RKM, Ho KY. Melatonin for the treatment of irritable bowel syndrome. World J Gastroenterol. 2014;20(10):2492-2498.
52. Garg P, Garg PK, Bhattacharya K. Psyllium husk positively alters gut microbiota, decreases inflammation, and has bowel-regulatory action, paving the way for physiologic management of irritable bowel syndrome. Gastroenterology. 2024;166(3):545-546.
 Anjali Pandit Bokota
Anjali Pandit Bokota Kristen Kimble
Kristen Kimble Bethany Doerfler
Bethany Doerfler Sara H. Marchese
Sara H. Marchese