With prevalence of type 2 diabetes and metabolic dysfunction-associated steatotic liver disease (MASLD) rising in parallel, there is an increased need for alternatives to surgical and pharmacological treatments for these conditions. Pharmacologic therapy can achieve good results in many patients, but for those with side effects, rising costs, or difficulty with adherence, the main alternative is bariatric surgery (which comes with significant cost and risk).17 Duodenal mucosal resurfacing (DMR) offers a less invasive alternative to bariatric surgery without the side effects and inconvenience of medication for patients with type 2 diabetes and MASLD.11
The duodenum is targeted in DMR because it is a major site of nutrient sensing and signaling in the gastrointestinal tract.2,3,4 The combination of bile acids, lipases, and incretin hormones help to control glucose levels in the blood and liver. Bariatric surgeries that bypass the duodenum or regulate the flow of nutrients into the duodenum show rapid improvements in glycemic control, making the duodenum a plausible site contributing to metabolic dysfunction.17
DMR is an endoscopic procedure that targets the superficial mucosa of the descending and inferior duodenum.8 The ablation of the mucosa is thought to reset the nutrient sensing and improve glycemic control.14 There is complete regrowth of the mucosa with minimal scarring and collagen deposition, suggesting the metabolic benefits are not structural.12
DMR is still under investigation, but early human feasibility studies and safety data show promising evidence. There have been consistent decreases in HbA1c and improved insulin sensitivity at follow-up, though these have not been studied long term.12 As MASLD is closely linked to insulin resistance and type 2 diabetes, DMR is being investigated as a potential adjunctive treatment with GLP-1 agonists and lifestyle modifications.15 Early evidence suggests that DMR has been associated with a decrease in liver lipid measurements, but this is still investigational.15 There are also multiple different technologies for DMR that are emerging, but have not been compared head-to-head.
Physiology and Pathophysiology of the Duodenum
Altered nutrient sensing pathways in the duodenum have been implicated in the development of insulin resistance and fatty liver disease, forming the basis for duodenal mucosal resurfacing as a targeted endoscopic intervention. Following a meal, there is a rise in the concentration of nutrients in the duodenum that are subsequently absorbed in the small bowel and released into the bloodstream.
Lipids are initially digested by lipases released by the stomach and pancreas, with a small contribution from salivary lipases. The presence of fatty acids and monoglycerides in the duodenum triggers the release of CCK from I-cells. CCK stimulates contraction of the gallbladder and release of bile acids into the duodenum. Bile acids help to emulsify the lipids, providing more surface area for lipases, and they also trigger the release of glucagon-like peptide-1 (GLP-1) from L-cells. GLP-1 helps to stimulate the release of insulin from the pancreas while also delaying gastric emptying and contributing to satiety, acting as a negative feedback mechanism.
Digestion of carbohydrates starts with salivary lipase, but this enzyme is quickly deactivated in stomach acid. The majority of carbohydrate digestion occurs in the small bowel via a combination of pancreatic amylase and brush border enzymes. Production and release of pancreatic amylase is stimulated by CCK. Starches and sugars are broken down into monosaccharides and are absorbed across enterocytes.
The main regulation of carbohydrate absorption is through nutrient sensing that adjusts the number of glucose transporters (SGLT-1 and GLUT-2) on the surface of enterocytes. The main nutrient sensors are T1R2 and T1R3 which are sweet taste receptors located on enterocytes and enteroendocrine cells. Activation of T1R2 and T1R3 increases GLUT-2 insertion into the apical membrane and SLGT-1 expression. In addition, it activates GLP-1 and GLP-2 levels to decrease the rate of gastric emptying. SLGT-1 also acts as a carbohydrate sensor that increases GLUT-2 and stimulates GLP-1 release.2
Systemic glucose levels are regulated through insulin and glucagon. Insulin is released following meals and it stimulates the uptake and storage of glucose. Glucagon opposes the action of insulin and is released during fasting to raise blood glucose levels.
In patients with type 2 diabetes and metabolic dysfunction-associated steatotic liver disease (MASLD) there is an exaggerated nutrient sensing response caused by mucosal hypertrophy, altered chemosensory receptor activity, and increased nutrient transporter expression. This impairs the regulation of glucose output from the liver and decreases sensitivity to insulin. These signaling changes are thought to contribute to the development and progression of type 2 diabetes and MASLD and form the basis for DMR. DMR aims to normalize these signaling pathways in the duodenum to augment the disease progression. DMR is thought to improve the signaling in the duodenum through ablating the tissue with dysfunctional signaling to act as a “reset” for the mucosa.
DMR Techniques, Devices, and Procedural Details
Hydrothermal DMR
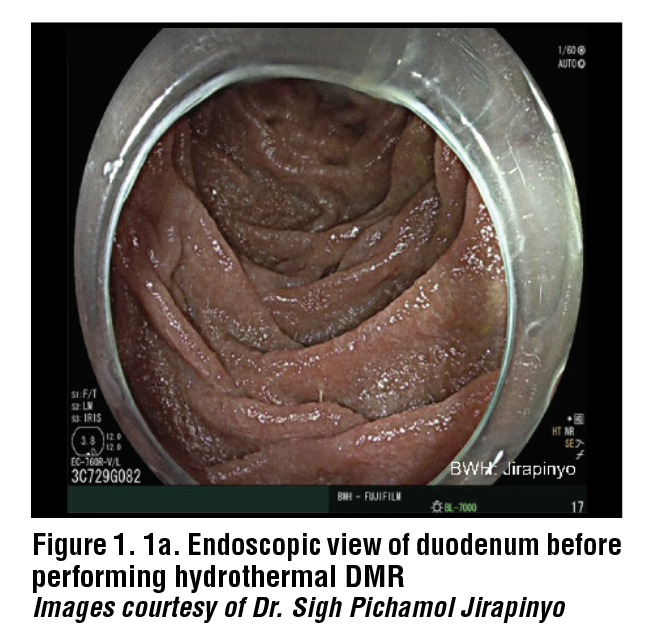
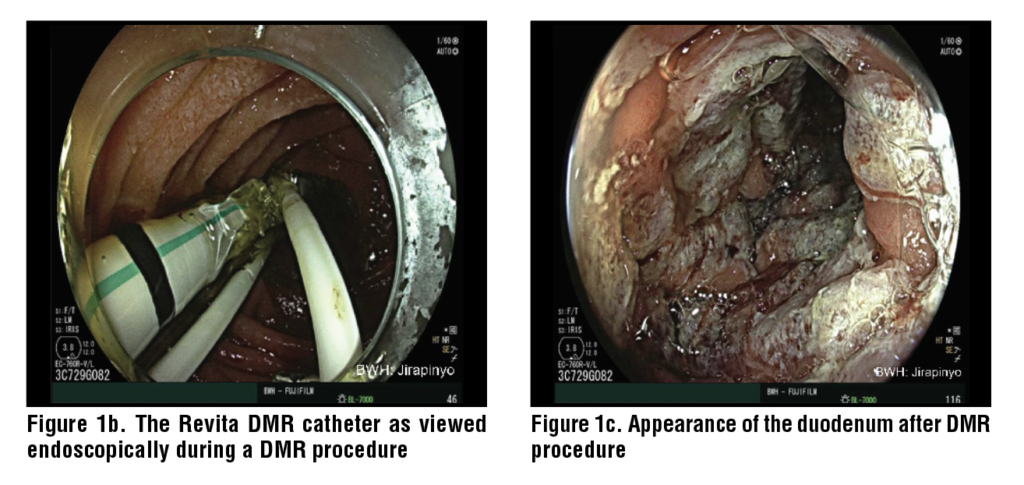
Hydrothermal DMR starts with the injection of a saline solution into the duodenal submucosa to lift the submucosa. This provides a barrier to prevent damage to the underlying muscularis propria and creates an even surface to ablate. Hydrothermal DMR uses a Revita catheter (Fractyl Health, Inc, Burlington, MA, USA) that has three submucosal injectors and a 2 cm balloon on the end. The balloon uses heated water to ablate the surface of the mucosa. The water inside the balloon reaches 80°C and ablates the surface of the mucosa for 10 seconds. The mucosa only requires one ablation before being advanced to the next segment of the duodenum until 10 cm of the duodenum has been treated. The ablation induces coagulative necrosis in the mucosal cells which form the most superficial 0.6mm of the duodenal wall. The muscularis propria lies at 1.0mm depth, and the ablation is not strong enough to disrupt that layer. The treatment is performed distal to the major duodenal papilla to ensure its safety. (Figure 1)
Prior to performing DMR, an upper endoscopy is required to evaluate the patient’s anatomy and exclude diseases of the mucosa such as duodenitis with ulcers, celiac disease, etc. Other conditions that may be contraindications to DMR include strictures, varices, and gastroduodenal ulcers, in addition to those previously mentioned. It is recommended that the wall of the duodenum be marked contralaterally to the major duodenal papilla to provide a proximal margin for treatment. A 0.035” guidewire should be passed to the proximal jejunum to act as a guide for the DMR catheter. Fluoroscopy can be utilized to ensure that the placement of the guidewire is correct. The Revita catheter can be placed over the guidewire and positioned just distal to the major duodenal papilla. For direct visualization during the procedure, an endoscope can be positioned in the duodenum just proximal to the balloon on the catheter. The gross appearance of the duodenum should be evaluated following the procedure to ensure the entire segment has been ablated. There are five ablation cycles to complete one treatment of 10cm of the duodenum.
Following the procedure, patients are generally discharged the same day or after an overnight hospitalization. Patients are prescribed a diet after the procedure that slowly advances from clear liquids to solid foods over two weeks during healing of the mucosa. After the two-week diet progression, patients should be counseled on important dietary changes that can help manage glucose control.
The ideal candidate for DMR is between age 28 and 75 with type 2 diabetes and a HbA1c between 7.5% and 10%, a body mass index (BMI) between 24-40 kg/m2 and preserved intrinsic insulin production. There are several contraindications for DMR including type 1 diabetes, low endogenous insulin production, previous gastrointestinal surgery that prohibits access to the duodenum, history of acute and chronic pancreatitis, history of duodenal inflammatory disease such as Crohn’s or Celiac disease, or anticoagulant therapy that cannot be discontinued. It is important that endogenous insulin production is maintained because the goal of DMR therapy is to improve insulin sensitivity, and those without endogenous insulin production may not benefit from the procedure.
Laser Duodenal Ablation
An emerging alternative to Hydrothermal DMR is Laser Duodenal Ablation using the Digma System (Digma Medical Ltd., Givat Shmuel, Israel). The Digma system consists of a control console and a single use catheter that is compatible with the working channel greater than 3.7 mm. The catheter uses a polyethylene terephthalate (PET) balloon that inflates to distend the duodenum. A continuous wave 5-15 W laser then delivers a focused beam to the duodenum, ablating 6 cm of the mucosa circumferentially per ablation cycle. The treatment starts at the duodenojejunal flexure and moves proximal, with up to seven ablations in three to four positions, ablating a total of 24 cm of the duodenal mucosa.
Irreversible Electroporation (IRE) Mucosal Ablation
Irreversible Electroporation (IRE) is an alternative to thermal ablation and uses pulsed electrical fields to create pores in cell membranes and induce apoptosis. The pulsed electrical fields of IRE also induce the renewal of the duodenal mucosa. The combination therapy is called recellularization via electroporation therapy (ReCET), performed using the Endogenex system (Endogenex Inc, Plymouth, Minnesota, USA). To perform ReCET, a guidewire is placed into the jejunum endoscopically. The position is confirmed using fluoroscopy and then the Endogenex catheter is introduced over the guidewire to the 2nd section of the duodenum. The ablation is performed proximally to distally, starting just distal to the major duodenal papilla. The catheter ablates 2 cm and two-thirds of the circumference of the duodenum at a time. The total segment of the duodenum that is treated is 10 cm.
Mechanism of Action
DMR ablates the mucosa and is thought to provide a reset for the signaling of enterocytes back to a healthier phenotype. This procedure allows for regrowth of the mucosa with minimal scarring and has shown significant decreases in HbA1c and blood glucose at follow-up.12 The mechanism of resulting decrease in HbA1c and blood glucose is uncertain, but is thought to be related to changes in enteroendocrine signaling. In follow-up histological evaluation, there was no change in the density of L and K cells after DMR, which are a key source of GLP-1 and GIP.13 This could mean that the mechanism of DMR alters hormonal signaling or cellular function rather than enteroendocrine cell number. There do not appear to be any structural changes to the mucosa, suggesting that the metabolic benefits of DMR are not mediated by structural damage or by inflammation.13
Western blot molecular analysis showed increased expression of PDZK1 and GATA6, which are involved in epithelial differentiation and nutrient signaling.13 This supports the hypothesis that DMR provides a reset for the duodenal mucosa which improves glucose homeostasis and downstream metabolic signaling.
Following DMR, there are significant decreases in insulin, glucagon and C-peptide levels in the blood. There are also significant decreases in postprandial glucose and glucagon concentrations.12
DMR is being investigated as a potential adjunctive therapy for non-alcoholic steatohepatitis (NASH) as there are limited treatment options beyond lifestyle modifications and pharmacologic therapy with agents such as GLP-1 agonists. Bariatric surgery is an option for some patients with NASH, but there are higher levels of surgical complications and morbidity in patients with cirrhosis, most of whom would be denied surgical therapy on these grounds. Current studies indicate there is improved glycemic control following DMR, but further studies are needed to show if there is a benefit in NASH.14
Combination Therapy with GLP-1 Receptor Agonists
With DMR acting locally to increase insulin levels in the blood and GLP-1 receptor agonists acting systemically to increase insulin secretion, the combination has potential to reduce insulin dependance and enhance glycemic control in patients with type 2 diabetes.2, 12 Early clinical studies have combined hydrothermal duodenal ablation with liraglutide or irreversible electroporation with semaglutide. All three studies included patients between ages 28 and 75 with type 2 diabetes on basal insulin with an HbA1c less than 8.0%.13,, The primary endpoint of all studies was the number of patients following DMR that were able to stay off of basal insulin with adequate glycemic control defined as an HbA1c of less than 7.5%. Of the 16 patients that underwent hydrothermal DMR with liraglutide, 11 of them (69%) had adequate control at 6 month follow up.16,17 At 18 month follow up, 8 patients (53%) remained off insulin therapy.16 A total of 14 patients underwent IRE with a range of electroporation doses from one 600V treatment to two 750V treatments.13 At 12 month follow up, 12 patients (86%) did not require insulin therapy, with the two patients needing basal insulin having only one 600V dose of electroporation.13
Safety and Adverse Events
Overall Safety Profile in Trials
DMR is generally well tolerated, with studies reporting mostly mild adverse events including nausea and abdominal pain following the procedure.7,10 In animal studies, there were no adverse events following the procedure, with only a mild inflammatory response noted in the duodenal mucosa. Histologically, the mucosal healing process was completed within six weeks. Some animals displayed increased collagen deposition in the submucosa of the treated region, but they displayed no functional limitations.7
In the first in human study of DMR with 29 patients, there were no adverse events reported during the procedure. Postprocedural adverse events were limited, with three patients out of the 29 experiencing duodenal stenosis following the ablation, that were treated using endoscopic dilation. The duodenal stenosis was determined to be caused by inadequate submucosal lift.7 There was complete regrowth of the mucosa observed at three months following the procedure in all 29 patients. There were 11 patients out of 29 that had evidence of low-to-intermediate fibrosis.7
In follow up clinic visits, there were transient and mild adverse events immediately following the procedure, most commonly abdominal pain, diarrhea, hyperglycemia, hypoglycemia, nasopharyngitis, and headache.10 On follow-up endoscopy 30 days after the procedure, the duodenum exhibited complete healing. After 30 days of healing, the most common adverse events were hypoglycemia and abdominal pain.10 There were no reported duodenal strictures and one instance of jejunal perforation that was caused by manipulation of the endoscope.10 There were also no clinical signs of pancreatitis, malabsorption, anemia, or infection.10
Long-Term Safety
Long-term safety profiles are still being developed, as DMR is an emerging/experimental procedure. After two years following DMR, trials indicate that the procedure was well tolerated with no device or procedure related serious adverse effects. Of the 46 patients that were followed, there were two reported adverse events that could have been linked to the procedure including one patient with constipation and one patient with general malaise and vitamin B12 deficiency.15 A larger sample size and a longer follow-up will be necessary, as data is very limited.
Comparative and Alternative Therapies
Bariatric Operations and Foregut Surgeries
Roux-en-Y gastric bypass and sleeve gastrectomy are bariatric surgical procedures that improve glycemic control and decrease weight in patients with type 2 diabetes and obesity. A Roux-en-Y gastric bypass is a restrictive and malabsorptive bariatric surgical procedure than involves bypassing the majority of the stomach and the whole duodenum to reduce food intake to promote weight loss., A sleeve gastrectomy involves removing 80% of the stomach along the greater curvature to promote early satiety and decrease the release of the hormone ghrelin that stimulates hunger signals.
Bariatric surgeries such as the Roux-en-Y gastric bypass and sleeve gastrectomy are indicated for people with a BMI of 35 kg/m2 or greater or a BMI of 30 kg/m2 with type 2 diabetes.18 Gastric bypass has been shown to provide long-term control of type 2 diabetes with decreased antidiabetic medicine use and higher rates of type 2 diabetes remission. There is a possibility of severe adverse events following gastric bypass surgery including bleeding, infection, and bowel leaks following the procedure. There can also be long-term issues afterwards such as dumping syndrome, nutritional deficiencies, gallstones, ulcers, and strictures.
While DMR aims to control blood glucose, Roux-en-Y gastric bypass and sleeve gastrectomy provide glycemic control while also controlling weight. Roux-en-Y gastric bypass causes greater weight loss than sleeve gastrectomy, but they provide similar improvements in glycemic control.16 Roux-en-Y gastric bypass can also help control symptoms of acid reflux. Both procedures encourage caloric restriction which aids in glycemic control, but they also alter nutrient flow, absorption, and incretin hormone release.16 Altered nutrient flow and incretin hormone release are also thought to be the basis for DMR.13
Pharmacologic Therapies
Pharmacologic therapies to provide glycemic control in patients with type 2 diabetes are alternatives or supplements to DMR that act through a variety of metabolic pathways. GLP-1 receptor agonists such as semaglutide and GLP-1/GIP agonists such as tirzepatide mimic the naturally occurring incretin hormone GLP-1 that is released from L-cells and GIP released from K-cells. Semaglutide and tirzepatide augment insulin and glucagon secretion while also slowing gastric emptying and decreasing caloric intake.
Slowed gastric emptying and decreased caloric intake result in decreased HbA1c and significant weight loss.22 These medications augment the same hormonal pathways that are hypothesized to decrease HbA1c levels in DMR.13 Both bariatric surgery and pharmacologic treatment cause weight loss, while weight changes following DMR have been modest and not always statistically significant.10, There is variability between cohorts and further studies would need to be completed to quantify the weight changes following DMR. It seems likely that, if widely implemented, DMR would often be used alongside GLP-1 agents.
Conclusion: Evidence Gaps, Limitations, and Research Priorities
As DMR is still an emerging and, to some extent, experimental procedure, clinical trial data is extremely limited. There is a need for trials in more patients to establish significant safety data for the procedure as well as examine the outcomes of a larger cohort. Trials have had varied inclusion criteria including HbA1c range, insulin use, and BMI. By standardizing the patient population, it allows for greater pooling of the data and comparison of results between trials.
Establishing long term safety and efficacy of DMR is difficult with trials for long term follow-up limited to 2 years and cohorts of less than 50 patients. Additional long-term follow-up data need to become available, and additional studies need to be done to increase the sample size.
The significant uncertainty about the mechanism of DMR needs to be investigated. By understanding the exact mechanism by which DMR improves metabolic outcomes, the procedure can be refined to be as least invasive as possible as well as more targeted. This can help determine the ideal candidate, the most effective treatment length as well as treatment frequency. Long term adverse events and metabolic changes need to be further elucidated.
Head-to-head comparisons between hydrothermal ablation and IRE have not been performed at this time but having trials of this type would help to clarify safety profiles of each modality. This could help direct further device development and provide future guidelines for treatment.
References
1. Musso G, Cassader M, Gambino R. Endoscopic duodenal mucosa
ablation: the future of diabetes treatment? Trends Mol Med. 2024
Jul;30(7):612-616. doi: 10.1016/j.molmed.2024.03.003. Epub
2024 Mar 29. PMID: 38553333.
2. Parikh A, Thevenin C. Physiology, Gastrointestinal Hormonal
Control. [Updated 2023 May 1]. In: StatPearls [Internet]. Treasure
Island (FL): StatPearls Publishing; 2025 Jan-. Available from:
https://www.ncbi.nlm.nih.gov/books/NBK537284/#
3. Rønnestad I, Akiba Y, Kaji I, Kaunitz JD. Duodenal luminal
nutrient sensing. Curr Opin Pharmacol. 2014 Dec;19:67-75. doi:
10.1016/j.coph.2014.07.010. Epub 2014 Aug 9. PMID: 25113991;
PMCID: PMC4845631.
4. Breen DM, Rasmussen BA, Côté CD, Jackson VM, Lam TK.
Nutrient-sensing mechanisms in the gut as therapeutic targets for
diabetes. Diabetes. 2013 Sep;62(9):3005-13. doi: 10.2337/db13-
0523. PMID: 23970519; PMCID: PMC3749331.
5. Holesh JE, Aslam S, Martin A. Physiology, Carbohydrates.
[Updated 2023 May 12]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK459280/#
6. Nakrani MN, Wineland RH, Anjum F. Physiology, Glucose
Metabolism. [Updated 2023 Jul 17]. In: StatPearls [Internet].
Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available
from: https://www.ncbi.nlm.nih.gov/books/NBK560599/
7. Habegger KM, Al-Massadi O, Heppner KM, Myronovych A,
Holland J, Berger J, Yi CX, Gao Y, Lehti M, Ottaway N, Amburgy
S, Raver C, Müller TD, Pfluger PT, Kohli R, Perez-Tilve D,
Seeley RJ, Tschöp MH. Duodenal nutrient exclusion improves
metabolic syndrome and stimulates villus hyperplasia. Gut. 2014
Aug;63(8):1238-46. doi: 10.1136/gutjnl-2013-304583. Epub 2013
Oct 9. PMID: 24107591; PMCID: PMC3981953.
8. Haidry RJ, van Baar AC, Galvao Neto MP, Rajagopalan H, Caplan
J, Levin PS, Bergman JJ, Rodriguez L, Deviere J, Thompson
CC. Duodenal mucosal resurfacing: proof-of-concept, procedural
development, and initial implementation in the clinical setting.
Gastrointest Endosc. 2019 Oct;90(4):673-681.e2. doi: 10.1016/j.
gie.2019.03.024. Epub 2019 Mar 29. PMID: 30935932
9. van Baar ACG, Haidry R, Rodriguez Grunert L, Galvao MPN,
Bisschops R, Hayee BH, Costamagna G, Deviere J, Bergman
JJGHM. Duodenal mucosal resurfacing: Multicenter experience
implementing a minimally invasive endoscopic procedure for
treatment of type 2 diabetes mellitus. Endosc Int Open. 2020
Nov;8(11):E1683-E1689. doi: 10.1055/a-1244-2283. Epub 2020
Oct 22. PMID: 33140025; PMCID: PMC7581486.
10. van Baar ACG, Holleman F, Crenier L, Haidry R, Magee C,
Hopkins D, Rodriguez Grunert L, Galvao Neto M, Vignolo
P, Hayee B, Mertens A, Bisschops R, Tijssen J, Nieuwdorp
M, Guidone C, Costamagna G, Devière J, Bergman JJGHM.
Endoscopic duodenal mucosal resurfacing for the treatment of
type 2 diabetes mellitus: one year results from the first international,
open-label, prospective, multicentre study. Gut. 2020
Feb;69(2):295-303. doi: 10.1136/gutjnl-2019-318349. Epub 2019
Jul 22. PMID: 31331994; PMCID: PMC6984054
11. Mingrone G, van Baar AC, Devière J, Hopkins D, Moura
E, Cercato C, Rajagopalan H, Lopez-Talavera JC, White K,
Bhambhani V, Costamagna G, Haidry R, Grecco E, Galvao Neto
M, Aithal G, Repici A, Hayee B, Haji A, Morris AJ, Bisschops
R, Chouhan MD, Sakai NS, Bhatt DL, Sanyal AJ, Bergman
JJGHM; Investigators of the REVITA-2 Study. Safety and efficacy
of hydrothermal duodenal mucosal resurfacing in patients with
type 2 diabetes: the randomised, double-blind, sham-controlled,
multicentre REVITA-2 feasibility trial. Gut. 2022 Feb;71(2):254-
264. doi: 10.1136/gutjnl-2020-323608. Epub 2021 Feb 17. PMID:
33597157; PMCID: PMC8761999.
12. Kovarova V, Lankova I, Machytka E, Knotkova K, Kratochvílová
H, Beneš M, Spicak J, Vasura A, Goldin E, Munter G, Zima
T, Mraz M, Dagan H, Levy B, Haluzik M, Kral J. Duodenal
Laser Ablation for Treatment of Type 2 Diabetes: Results of
First in Human Study. United European Gastroenterol J. 2025
Jun;13(5):750-758. doi: 10.1002/ueg2.12762. Epub 2025 Feb 20.
PMID: 39976927; PMCID: PMC12188360.
13. Busch CBE, Meiring S, van Baar ACG, Holleman F, Nieuwdorp
M, Bergman JJGHM. Recellularization via electroporation therapy
of the duodenum combined with glucagon-like peptide-1 receptor
agonist to replace insulin therapy in patients with type 2 diabetes:
12-month results of a first-in-human study. Gastrointest Endosc.
2024 Nov;100(5):896-904. doi: 10.1016/j.gie.2024.04.2904. Epub
2024 Apr 29. PMID: 38692517.
14. Meiring S, Aydin Ö, van Baar ACG, van der Vossen EWJ,
Rampanelli E, van Grieken NCT, Holleman F, Nieuwdorp
M, Bergman JJGHM. From Endoscopic Inspection to Gene-
Expression: A Thorough Assessment of the Duodenal Mucosa
After Resurfacing-A Prospective Study. Dig Dis Sci. 2025
Mar;70(3):1052-1063. doi: 10.1007/s10620-024-08710-4. Epub
2025 Jan 8. PMID: 39779586; PMCID: PMC11920325.
15. Chuang TJ, Ko CW, Shiu SI. The metabolic influence of duodenal
mucosal resurfacing for nonalcoholic fatty liver disease.
Medicine (Baltimore). 2023 Oct 6;102(40):e35147. doi:
10.1097/MD.0000000000035147. PMID: 37800801; PMCID:
PMC10553053.
16. Meiring S, Busch CBE, van Baar ACG, Hemke R, Holleman
F, Nieuwdorp M, Bergman JJGHM. Eliminating exogenous
insulin therapy in patients with type 2 diabetes by duodenal
ablation and GLP-1RA decreases risk scores for cardiovascular
events. Cardiovasc Diabetol. 2022 Sep 22;21(1):191. doi: 10.1186/
s12933-022-01628-z. PMID: 36138441; PMCID: PMC9503196.
17. van Baar ACG, Meiring S, Smeele P, Vriend T, Holleman F,
Barlag M, Mostafavi N, Tijssen JGP, Soeters MR, Nieuwdorp M,
Bergman JJGHM. Duodenal mucosal resurfacing combined with
glucagon-like peptide-1 receptor agonism to discontinue insulin
in type 2 diabetes: a feasibility study. Gastrointest Endosc. 2021
Jul;94(1):111-120.e3. doi: 10.1016/j.gie.2020.12.021. Epub 2020
Dec 24. PMID: 33359437.
18. van Baar ACG, Devière J, Hopkins D, Crenier L, Holleman F,
Galvão Neto MP, Becerra P, Vignolo P, Rodriguez Grunert L,
Mingrone G, Costamagna G, Nieuwdorp M, Guidone C, Haidry RJ,
Hayee B, Magee C, Carlos Lopez-Talavera J, White K, Bhambhani
V, Cozzi E, Rajagopalan H, J G H M Bergman J. Durable metabolic
improvements 2 years after duodenal mucosal resurfacing
(DMR) in patients with type 2 diabetes (REVITA-1 Study).
Diabetes Res Clin Pract. 2022 Feb;184:109194. doi: 10.1016/j.
diabres.2022.109194. Epub 2022 Jan 13. PMID: 35032562.
19. Wallenius V, Dirinck E, Fändriks L, Maleckas A, le Roux CW,
Thorell A. Glycemic Control after Sleeve Gastrectomy and Roux-
En-Y Gastric Bypass in Obese Subjects with Type 2 Diabetes
Mellitus. Obes Surg. 2018 Jun;28(6):1461-1472. doi: 10.1007/
s11695-017-3061-3. PMID: 29264780; PMCID: PMC5973990.
20. Umeda LM, Silva EA, Carneiro G, Arasaki CH, Geloneze B,
Zanella MT. Early improvement in glycemic control after bariatric
surgery and its relationships with insulin, GLP-1, and glucagon
secretion in type 2 diabetic patients. Obes Surg 2011;21:896–901
21. Mitchell BG, Collier SA, Gupta N. Roux-en-Y Gastric Bypass.
[Updated 2024 Nov 9]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK553157/
22. Seeras K, Sankararaman S, Lopez PP. Sleeve Gastrectomy.
[Updated 2023 Jan 23]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK519035/
23. Pories WJ, Swanson MS, MacDonald KG, Long SB, Morris PG,
Brown BM, Barakat HA, deRamon RA, Israel G, Dolezal JM, et
al. Who would have thought it? An operation proves to be the most
effective therapy for adult-onset diabetes mellitus. Ann Surg. 1995
Sep;222(3):339-50; discussion 350-2. doi: 10.1097/00000658-
199509000-00011. PMID: 7677463; PMCID: PMC1234815.
24. Seeras K, Acho RJ, Lopez PP. Roux-en-Y Gastric Bypass Chronic
Complications. [Updated 2023 Jun 5]. In: StatPearls [Internet].
Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available
from: https://www.ncbi.nlm.nih.gov/books/NBK519489/#
25. Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists
in the treatment of type 2 diabetes – state-of-the-art. Mol
Metab. 2021 Apr;46:101102. doi: 10.1016/j.molmet.2020.101102.
Epub 2020 Oct 14. PMID: 33068776; PMCID: PMC8085572.
26. Rajagopalan H, Cherrington AD, Thompson CC, Kaplan LM,
Rubino F, Mingrone G, Becerra P, Rodriguez P, Vignolo P, Caplan
J, Rodriguez L, Galvao Neto MP. Endoscopic Duodenal Mucosal
Resurfacing for the Treatment of Type 2 Diabetes: 6-Month
Interim Analysis From the First-in-Human Proof-of-Concept
Study. Diabetes Care. 2016 Dec;39(12):2254-2261. doi: 10.2337/
dc16-0383. Epub 2016 Aug 12. PMID: 27519448.
 Moriah Glennon
Moriah Glennon Douglas G. Adler
Douglas G. Adler