Women with Inflammatory Bowel Disease (IBD) increasingly utilize assisted reproductive technologies (ART), such as in vitro fertilization (IVF) and oocyte cryopreservation. Women with active IBD or a history of pelvic surgery may have higher rates of infertility compared to the general population. Currently, data regarding ART and induction of IBD flare are limited. However, recent evidence demonstrates that ART is safe in women with well-controlled IBD, with the rate of disease flare during ART procedures remaining low. This article reviews the current evidence on fertility in IBD, when to refer patients for evaluation, safety and efficacy of ART, and cost considerations. Physicians should counsel patients that achieving disease remission prior to conception is strongly recommended and reassure patients that most IBD medications do not appear to impair the efficacy of ART. With a multidisciplinary approach, we can improve fertility rates and pregnancy outcomes among women with IBD.
Introduction
The incidence of IBD, comprising Crohn’s disease (CD) and Ulcerative Colitis (UC), peaks between the ages of 15–30 years across Western Europe and North America, with a second smaller peak occurring between the ages of 60–79 years. As 25% of all IBD patients are diagnosed before the age of twenty, the disease burden is often concentrated during a woman’s reproductive years.1
Caring for a young woman with IBD often involves addressing concerns regarding medication safety and risk of flares during pregnancy. Women with IBD face higher rates of voluntary childlessness compared to the general population, a phenomenon caused by misconceptions rather than physical inability.2,4
This article addresses practical questions providers will face behind closed doors. Could I ever have children? Should I see a fertility specialist? Is IVF safe for me? Will medications hurt my baby? Will I flare during IVF treatment? By using up-to-date literature, we offer a roadmap for optimizing reproductive health in this population.
Understanding Fertility in IBD
It is a common misconception that a diagnosis of IBD means infertility. Up to 18% of women with CD and 14% women with UC may experience infertility.12 However, this increased risk of infertility is not generalizable across all women with IBD. In fact, multiple systematic reviews and national cohort studies suggest there are similar fertility rates in women with UC and slightly decreased fertility rates in women with CD compared to the general population.2 The primary drivers of reduced fertility are active disease at the time of conception, prior pelvic surgery (particularly ileal pouch-anal anastomosis), and active peri-anal disease.1,2 Decreased fertility is most often seen after ileal-pouch-anal anastomosis (J-pouch) surgery from pelvic adhesions and tubal obstruction while active disease can lead to a dysfunctional placenta and reduced ovarian reserve, reflected by decreased anti-Mullerian hormone levels. Fortunately, systematic reviews have reported that women with IBD in remission have similar ovarian reserves to healthy controls.2,10 This data supports the key clinical target of maintaining 3-6 months of remission prior to conception.
Table 1. Myths and Facts About IBD And Pregnancy
| MYTHS | FACTS |
| If I have IBD, my child will get it | While genetics can play a role, the absolute risk to a child is lower than expected. If one parent has IBD, the risk of a child developing the disease is around 6-9% (compared to ~1% in the general population). If both parents have IBD, the risk increases to 30%.1,2 |
| There is nothing I can do to lower the risk for my child getting IBD | Environmental exposures during pregnancy may play a role in IBD pathogenesis, and many are modifiable. Smoking: Maternal smoking is associated with a 1.5-fold increased risk of IBD in the offspring. Smoking cessation is the single most effective action a patient can take.2,3 Antibiotics: Prenatal exposure to antibiotics, especially multiple courses or use during the third trimester, is associated with increased risk of IBD in the offspring, likely due to maternal microbiome disruption. While antibiotics should be used for necessary indications, antibiotic stewardship is encouraged.1,2 Diet and Food Additives: The MOMMY-IBD study, a prospective birth cohort from China, found that mothers with IBD consumed higher amounts of food additives compared to non-IBD mothers. Higher intake of food additives was associated with depletion of beneficial gut Bacteroides species and proliferation of Streptococcus species. Fecal calprotectin levels were significantly higher in infants born to mothers with higher food additive intake, regardless of parental IBD status. Common dietary emulsifiers have been shown to directly increase pro-inflammatory potential and promote dysbiosis. Encouraging a diet rich in natural, whole foods while limiting ultra-processed foods containing emulsifiers and additives may support the development of a healthy infant microbiome and potentially reduce IBD risk in offspring.17 |
| I need to stop my IBD medications before getting pregnant | Discontinuing effective therapy is rarely necessary and often dangerous. The risk of disease flare poses a far greater risk of poor outcomes to both the pregnancy and the fetus than the medications themselves.2 Maintenance therapies, including 5-ASAs and biologics, should be continued throughout gestation. Thiopurines are also considered low risk, as current data show no increased risk of congenital malformations.1,2,4 The only exception is methotrexate (stop 1-3 months prior to conception). JAK inhibitors (tofacitinib, upadacitinib, filgotinib), and S1P modulators should be discontinued 4 weeks prior to attempting conception unless there is no other viable option for the mother.1,2,13 |
| IBD medications will cause birth defects | Multiple systematic reviews, including the PIANO registry covering > 1,400 live births, found no increased risk of congenital malformations associated with anti-TNFs, thiopurines, or combination therapy. Biologics consist of large monoclonal antibodies that cannot cross the placenta passively during first trimester during key fetal organogenesis (weeks 2-8), making teratogenicity unlikely. Most monoclonal antibody transfers actively in the third trimester (~80%). However, small molecules (JAK inhibitors, S1P receptor modulators) can cross the placenta throughout pregnancy, raising concerns about potential teratogenicity.2 Overall, the rate of congenital anomalies in exposed infants is similar to the general population. Conversely, while corticosteroids are not teratogenic, they increase the risk of preterm birth and gestational diabetes; therefore, providers should strive to get patients on steroid-sparing agents and into remission.1,2,3,5 |
| I cannot breastfeed while on IBD medications | Breastfeeding is safe and encouraged for able mothers with IBD. Monoclonal antibodies (biologics) have very poor transfer into breast milk, with less than 1% of maternal serum levels.2 Any amount ingested is largely broken down by the infant’s digestive system before it can be absorbed.1,2 Clinical data from the PIANO registry confirms that breastfed infants exposed to biologics meet developmental milestones at the same rates as unexposed or non-breastfed infants and do not have an increased risk of infection or other complications.1,2 While global guidelines advocate for breastfeeding for at least 6-12 months, a clear distinction must be made for small molecule therapies. Women treated with methotrexate, cyclosporine, allopurinol, JAK inhibitors, and S1P modulators should avoid breastfeeding, as these smaller compounds can readily cross into breast milk.2 |
| All women with IBD need a C-section | An IBD diagnosis does not always require a C-section. For many patients, vaginal delivery is safe and appropriate.2,5 C-sections are recommended in women with active perianal Crohn’s disease (including complex perianal fistulas, rectovaginal fistulas, active rectal inflammation, rectal abscesses) and a history of ileal pouch-anal anastomosis (IPAA).1,2,5 |
| My baby cannot receive vaccines if I was on biologics during pregnancy | Infants exposed to biologics in utero can and should receive most vaccinations on schedule. The Bacillus Calmette-Guérin (BCG) vaccine and live oral polio (U.S. uses inactive polio vaccine) should be avoided for the first 6 months of life among infants exposed to biologics in utero.2 New data indicate that the live rotavirus vaccination is safe in this population, with studies showing no serious adverse events in infants with in utero biologic exposure.2 Regarding breastfeeding, there should be minimal to no transfer of biologic medications into the breastmilk, and thus, do not cause systemic immunosuppression in the infant. Live vaccines should therefore be given on the standard schedule regardless of breastfeeding.1,2 |
| I cannot take aspirin during pregnancy because it will cause an IBD flare | While IBD patients are typically counseled to avoid NSAIDs to prevent disease flare, low-dose aspirin is an exception. There is no evidence of increased IBD disease activity while on low-dose aspirin (150–162 mg). Recent studies have confirmed that flare rates were similar between women taking low-dose aspirin compared to those who were not.1,2,5 Low-dose aspirin can reduce the risk of preterm preeclampsia by over 60%. Because women with IBD are at increased risk for preeclampsia, it is recommended to initiate low-dose aspirin (~150-162 mg) between 12 and 16 weeks of gestation and continue throughout pregnancy.2 |
For gastroenterologists, it is also important to screen for non-biological causes of infertility. Decreased libido and dyspareunia resulting from active perianal or pelvic disease are frequently overlooked. Additionally, misinformation regarding medication teratogenicity, heritability, and poor pregnancy outcomes also contributes to voluntary infertility.2 A review of sexual function and common misconceptions is the best first step in identifying patients who need gastroenterology or fertility support.
Combating Misinformation
Patients often rely on unverified online sources for health information. Gastroenterologists can use the evidence-based talking points in Table 1 to address some common myths. Additionally, they can refer them to the PIANOstudy.org website for a patient education video in seven languages.
When to Refer for Fertility Evaluation
Gastroenterologists should have a lower threshold for referral to a reproductive endocrinologist in patients with IBD compared to the general population. While standard guidelines recommend referral after 12 months of unsuccessful conception, women with IBD, particularly those with CD or prior pelvic surgery, should be referred after 6 months of trying. For patients older than 40 years or those with extensive pelvic surgeries (such as J-pouch), referral after 4 months of unsuccessful conception is appropriate.3,4
Early referral to a fertility expert is important for three reasons:
Anatomical complications from prior surgeries may create a challenging pathway to natural conception.3
If a patient has active disease, the path to remission may require medication changes and imaging/endoscopic evaluation, a process that can take several months.2,3
Natural age-related fertility decline compounds the existing fertility challenges in those with chronic inflammatory conditions such as IBD.2,3,5
Preconception/Pre-ART Optimization
All women with IBD of childbearing age should be offered pre-conception counseling. The goal should be made clear: endoscopic and steroid-free clinical remission for 3-6 months prior to pursuing ART or natural conception.2,5
The rationale for this recommendation is that disease activity within six months of conception is associated with a 5-fold increased risk of disease activity during pregnancy. Active IBD increases the risk of adverse outcomes, including pre-term birth, low fetal birth weight, pre-eclampsia, and C-section delivery.2,4,5
Confirmation of remission is more than the lack of clinical symptoms. Gastroenterologists should assess for:
Fecal calprotectin <150 μg/g
Normal C-Reactive Protein (CRP)
Mucosal healing on colonoscopy or flexible sigmoidoscopy or transmural healing on intestinal ultrasound
Adequate drug levels for thiopurines and anti-TNFs
If disease activity is present, the patient should ideally optimize therapy and delay conception until optimal control is achieved.2 For those pursuing ART, remission is also recommended to optimize tolerance of hormonal therapies and maximize the success of implantation.
Pre-conception/Pre-ART Management
The safety of IBD medication during pregnancy planning is a frequent source of confusion. The current data suggest that most IBD medications have no negative effects on egg harvesting, ART efficacy, or pregnancy rates.
1. Safe to continue: Anti-TNFs, 5-ASA (mesalamine), thiopurines (azathioprine), corticosteroids, integrin blockers (vedolizumab), and IL-12/23 and IL-23 inhibitors (ustekinumab, risankizumab, mirikizumab, guselkumab).2,13
2. Must discontinue:
a. Methotrexate: A known teratogen and abortifacient, this should be discontinued at least 1 month prior to attempting conception.1,2,13
b. Small Molecules: JAK inhibitors (e.g., upadacitinib, tofacitinib) and S1P receptor modulators (e.g., ozanimod) should be discontinued at least 4 weeks prior to conception due to limited safety data unless there is no other viable option for the mother.1,13 While there is growing, but small, data on use in pregnancy, there is no data on use during cryopreservation. However, there is currently no evidence or theoretical reason to think a risk may exist. A risk-to-benefit discussion should be had with the patient on these medications. In many cases, given disease severity, the drug is continued.
Safety and Efficacy of ART in IBD
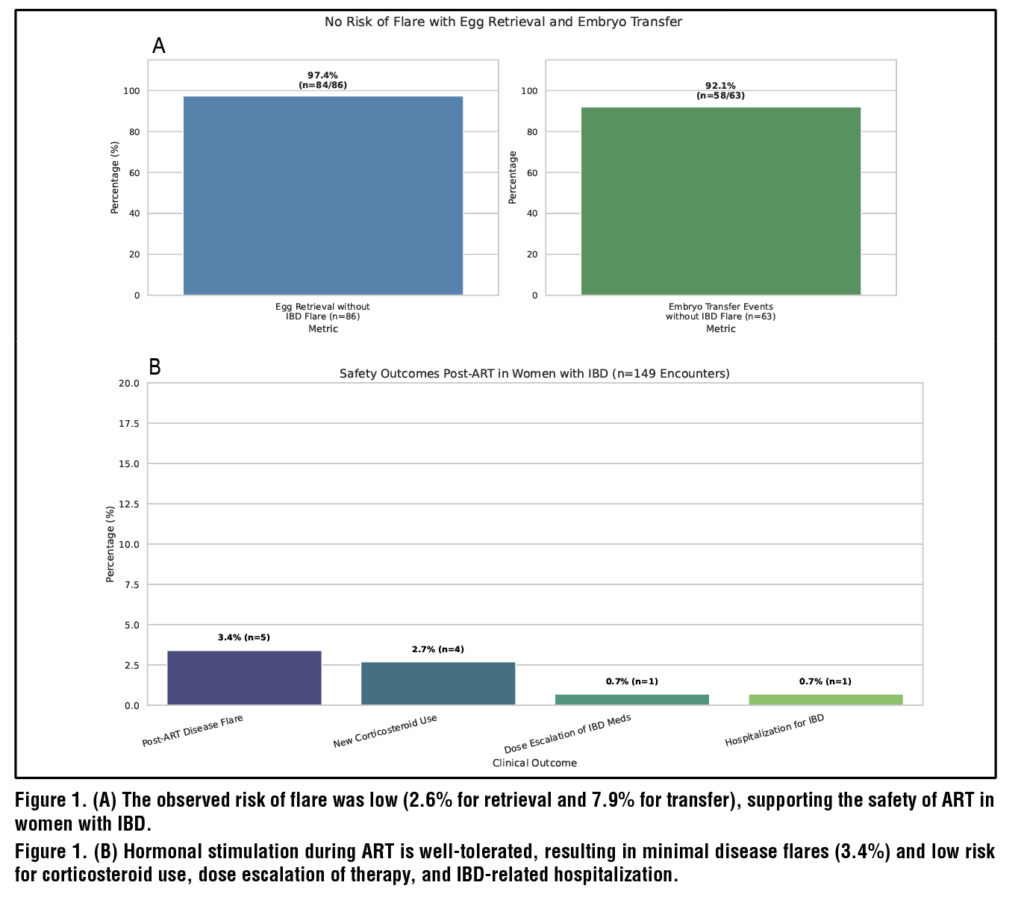
Flares occurred in 3.4% of post-ART encounters with minimal IBD-related hospitalization (0.7%), steroid use post-ART (2.7%), and medication escalation (1.3%). (Figure 1B)
Efficacy was also high, with egg retrieval rates exceeding 97% and an embryo transfer rate of approximately 92% occurring without an IBD flare. (Figure 1A)
Multiple cohort studies confirm these findings, showing that women with medically managed IBD achieve live birth rates comparable to healthy controls.8 These findings suggest that ART is low risk for flare among women with IBD and is effective.8,11,14
However, those with prior surgeries have different outcomes. Women with a J-pouch have a 64% lower live birth rate after IVF compared to those who have their UC medically managed. Similarly, women with CD who have had prior pelvic surgeries have a 49%-71% lower live birth rate after ART compared to those with medically managed CD.3
Options and Financial Considerations
Women with IBD should be aware of the many ways to have a family – natural conception, ART, surrogacy, and adoption. For those with significant prior or current disease burden or with medication concerns, these options should be discussed. For women undergoing colectomy for UC, many centers offer the option of a subtotal proctocolectomy with ileostomy and rectal stump during childbearing to avoid scarring in the pelvis.
The financial burden of surrogacy and ART represents a significant barrier for many patients struggling to conceive naturally. The average cost of one cycle of IVF ranges from $15,000-$30,000 when medications are used.6,16 Since many patients require multiple cycles to achieve pregnancy, the costs can quickly escalate. A financing industry survey noted that 70% of women who underwent IVF went into debt, and 34% of respondents reported cessation of treatment due to high treatment costs.15
Insurance coverage for ART is not standardized. As of 2025, 25 states have some form of fertility insurance by law, but coverage often includes spending caps or cycle limits. Patients should understand their insurance benefits and plan accordingly. Financial stress can add to the emotional toll of fertility treatment.15,16
Summary
- Fertility among women with IBD in remission is similar to the general population. Infertility is usually driven by active disease or prior pelvic surgery.
- Misinformation often drives voluntary childlessness. Reassure patients that the risk of passing IBD to a child is low and that most medications are safe to use.
- Stop methotrexate during conception, pregnancy, and breastfeeding.
- JAK inhibitors and S1P modulators should be stopped unless there is no viable option for maternal health
- Continue biologics and thiopurines.
- Referral to a fertility expert should be made early – after 6 months of concerted attempts or 4 months if the patient is older than 40 and/or has had prior pelvic surgery.
- Pre-conception optimization requires 3-6 months of steroid-free remission. Active disease at conception increases the risk of flares by 5-fold and increases risk for pre-term birth, low birth weight, C-section deliveries, and NICU admission.
- ART is safe in IBD patients, with low rates of flares (~3%), steroid use (2.7%), and hospitalizations (<1%).
- Discuss the various options to have a family and prepare patients for the high cost of IVF ($15,000-$30,000 per cycle) with variable insurance coverage.
- With proper counseling and multidisciplinary support, we can even the scales, giving women with IBD a safe path to a successful pregnancy and a healthy family.
References
1. Musso G, Cassader M, Gambino R. Endoscopic duodenal mucosa
ablation: the future of diabetes treatment? Trends Mol Med. 2024
Jul;30(7):612-616. doi: 10.1016/j.molmed.2024.03.003. Epub
2024 Mar 29. PMID: 38553333.
2. Parikh A, Thevenin C. Physiology, Gastrointestinal Hormonal
Control. [Updated 2023 May 1]. In: StatPearls [Internet]. Treasure
Island (FL): StatPearls Publishing; 2025 Jan-. Available from:
https://www.ncbi.nlm.nih.gov/books/NBK537284/#
3. Rønnestad I, Akiba Y, Kaji I, Kaunitz JD. Duodenal luminal
nutrient sensing. Curr Opin Pharmacol. 2014 Dec;19:67-75. doi:
10.1016/j.coph.2014.07.010. Epub 2014 Aug 9. PMID: 25113991;
PMCID: PMC4845631.
4. Breen DM, Rasmussen BA, Côté CD, Jackson VM, Lam TK.
Nutrient-sensing mechanisms in the gut as therapeutic targets for
diabetes. Diabetes. 2013 Sep;62(9):3005-13. doi: 10.2337/db13-
0523. PMID: 23970519; PMCID: PMC3749331.
5. Holesh JE, Aslam S, Martin A. Physiology, Carbohydrates.
[Updated 2023 May 12]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK459280/#
6. Nakrani MN, Wineland RH, Anjum F. Physiology, Glucose
Metabolism. [Updated 2023 Jul 17]. In: StatPearls [Internet].
Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available
from: https://www.ncbi.nlm.nih.gov/books/NBK560599/
7. Habegger KM, Al-Massadi O, Heppner KM, Myronovych A,
Holland J, Berger J, Yi CX, Gao Y, Lehti M, Ottaway N, Amburgy
S, Raver C, Müller TD, Pfluger PT, Kohli R, Perez-Tilve D,
Seeley RJ, Tschöp MH. Duodenal nutrient exclusion improves
metabolic syndrome and stimulates villus hyperplasia. Gut. 2014
Aug;63(8):1238-46. doi: 10.1136/gutjnl-2013-304583. Epub 2013
Oct 9. PMID: 24107591; PMCID: PMC3981953.
8. Haidry RJ, van Baar AC, Galvao Neto MP, Rajagopalan H, Caplan
J, Levin PS, Bergman JJ, Rodriguez L, Deviere J, Thompson
CC. Duodenal mucosal resurfacing: proof-of-concept, procedural
development, and initial implementation in the clinical setting.
Gastrointest Endosc. 2019 Oct;90(4):673-681.e2. doi: 10.1016/j.
gie.2019.03.024. Epub 2019 Mar 29. PMID: 30935932
9. van Baar ACG, Haidry R, Rodriguez Grunert L, Galvao MPN,
Bisschops R, Hayee BH, Costamagna G, Deviere J, Bergman
JJGHM. Duodenal mucosal resurfacing: Multicenter experience
implementing a minimally invasive endoscopic procedure for
treatment of type 2 diabetes mellitus. Endosc Int Open. 2020
Nov;8(11):E1683-E1689. doi: 10.1055/a-1244-2283. Epub 2020
Oct 22. PMID: 33140025; PMCID: PMC7581486.
10. van Baar ACG, Holleman F, Crenier L, Haidry R, Magee C,
Hopkins D, Rodriguez Grunert L, Galvao Neto M, Vignolo
P, Hayee B, Mertens A, Bisschops R, Tijssen J, Nieuwdorp
M, Guidone C, Costamagna G, Devière J, Bergman JJGHM.
Endoscopic duodenal mucosal resurfacing for the treatment of
type 2 diabetes mellitus: one year results from the first international,
open-label, prospective, multicentre study. Gut. 2020
Feb;69(2):295-303. doi: 10.1136/gutjnl-2019-318349. Epub 2019
Jul 22. PMID: 31331994; PMCID: PMC6984054
11. Mingrone G, van Baar AC, Devière J, Hopkins D, Moura
E, Cercato C, Rajagopalan H, Lopez-Talavera JC, White K,
Bhambhani V, Costamagna G, Haidry R, Grecco E, Galvao Neto
M, Aithal G, Repici A, Hayee B, Haji A, Morris AJ, Bisschops
R, Chouhan MD, Sakai NS, Bhatt DL, Sanyal AJ, Bergman
JJGHM; Investigators of the REVITA-2 Study. Safety and efficacy
of hydrothermal duodenal mucosal resurfacing in patients with
type 2 diabetes: the randomised, double-blind, sham-controlled,
multicentre REVITA-2 feasibility trial. Gut. 2022 Feb;71(2):254-
264. doi: 10.1136/gutjnl-2020-323608. Epub 2021 Feb 17. PMID:
33597157; PMCID: PMC8761999.
12. Kovarova V, Lankova I, Machytka E, Knotkova K, Kratochvílová
H, Beneš M, Spicak J, Vasura A, Goldin E, Munter G, Zima
T, Mraz M, Dagan H, Levy B, Haluzik M, Kral J. Duodenal
Laser Ablation for Treatment of Type 2 Diabetes: Results of
First in Human Study. United European Gastroenterol J. 2025
Jun;13(5):750-758. doi: 10.1002/ueg2.12762. Epub 2025 Feb 20.
PMID: 39976927; PMCID: PMC12188360.
13. Busch CBE, Meiring S, van Baar ACG, Holleman F, Nieuwdorp
M, Bergman JJGHM. Recellularization via electroporation therapy
of the duodenum combined with glucagon-like peptide-1 receptor
agonist to replace insulin therapy in patients with type 2 diabetes:
12-month results of a first-in-human study. Gastrointest Endosc.
2024 Nov;100(5):896-904. doi: 10.1016/j.gie.2024.04.2904. Epub
2024 Apr 29. PMID: 38692517.
14. Meiring S, Aydin Ö, van Baar ACG, van der Vossen EWJ,
Rampanelli E, van Grieken NCT, Holleman F, Nieuwdorp
M, Bergman JJGHM. From Endoscopic Inspection to Gene-
Expression: A Thorough Assessment of the Duodenal Mucosa
After Resurfacing-A Prospective Study. Dig Dis Sci. 2025
Mar;70(3):1052-1063. doi: 10.1007/s10620-024-08710-4. Epub
2025 Jan 8. PMID: 39779586; PMCID: PMC11920325.
15. Chuang TJ, Ko CW, Shiu SI. The metabolic influence of duodenal
mucosal resurfacing for nonalcoholic fatty liver disease.
Medicine (Baltimore). 2023 Oct 6;102(40):e35147. doi:
10.1097/MD.0000000000035147. PMID: 37800801; PMCID:
PMC10553053.
16. Meiring S, Busch CBE, van Baar ACG, Hemke R, Holleman
F, Nieuwdorp M, Bergman JJGHM. Eliminating exogenous
insulin therapy in patients with type 2 diabetes by duodenal
ablation and GLP-1RA decreases risk scores for cardiovascular
events. Cardiovasc Diabetol. 2022 Sep 22;21(1):191. doi: 10.1186/
s12933-022-01628-z. PMID: 36138441; PMCID: PMC9503196.
17. van Baar ACG, Meiring S, Smeele P, Vriend T, Holleman F,
Barlag M, Mostafavi N, Tijssen JGP, Soeters MR, Nieuwdorp M,
Bergman JJGHM. Duodenal mucosal resurfacing combined with
glucagon-like peptide-1 receptor agonism to discontinue insulin
in type 2 diabetes: a feasibility study. Gastrointest Endosc. 2021
Jul;94(1):111-120.e3. doi: 10.1016/j.gie.2020.12.021. Epub 2020
Dec 24. PMID: 33359437.
18. van Baar ACG, Devière J, Hopkins D, Crenier L, Holleman F,
Galvão Neto MP, Becerra P, Vignolo P, Rodriguez Grunert L,
Mingrone G, Costamagna G, Nieuwdorp M, Guidone C, Haidry RJ,
Hayee B, Magee C, Carlos Lopez-Talavera J, White K, Bhambhani
V, Cozzi E, Rajagopalan H, J G H M Bergman J. Durable metabolic
improvements 2 years after duodenal mucosal resurfacing
(DMR) in patients with type 2 diabetes (REVITA-1 Study).
Diabetes Res Clin Pract. 2022 Feb;184:109194. doi: 10.1016/j.
diabres.2022.109194. Epub 2022 Jan 13. PMID: 35032562.
19. Wallenius V, Dirinck E, Fändriks L, Maleckas A, le Roux CW,
Thorell A. Glycemic Control after Sleeve Gastrectomy and Roux-
En-Y Gastric Bypass in Obese Subjects with Type 2 Diabetes
Mellitus. Obes Surg. 2018 Jun;28(6):1461-1472. doi: 10.1007/
s11695-017-3061-3. PMID: 29264780; PMCID: PMC5973990.
20. Umeda LM, Silva EA, Carneiro G, Arasaki CH, Geloneze B,
Zanella MT. Early improvement in glycemic control after bariatric
surgery and its relationships with insulin, GLP-1, and glucagon
secretion in type 2 diabetic patients. Obes Surg 2011;21:896–901
21. Mitchell BG, Collier SA, Gupta N. Roux-en-Y Gastric Bypass.
[Updated 2024 Nov 9]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK553157/
22. Seeras K, Sankararaman S, Lopez PP. Sleeve Gastrectomy.
[Updated 2023 Jan 23]. In: StatPearls [Internet]. Treasure Island
(FL): StatPearls Publishing; 2025 Jan-. Available from: https://
www.ncbi.nlm.nih.gov/books/NBK519035/
23. Pories WJ, Swanson MS, MacDonald KG, Long SB, Morris PG,
Brown BM, Barakat HA, deRamon RA, Israel G, Dolezal JM, et
al. Who would have thought it? An operation proves to be the most
effective therapy for adult-onset diabetes mellitus. Ann Surg. 1995
Sep;222(3):339-50; discussion 350-2. doi: 10.1097/00000658-
199509000-00011. PMID: 7677463; PMCID: PMC1234815.
24. Seeras K, Acho RJ, Lopez PP. Roux-en-Y Gastric Bypass Chronic
Complications. [Updated 2023 Jun 5]. In: StatPearls [Internet].
Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available
from: https://www.ncbi.nlm.nih.gov/books/NBK519489/#
25. Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists
in the treatment of type 2 diabetes – state-of-the-art. Mol
Metab. 2021 Apr;46:101102. doi: 10.1016/j.molmet.2020.101102.
Epub 2020 Oct 14. PMID: 33068776; PMCID: PMC8085572.
26. Rajagopalan H, Cherrington AD, Thompson CC, Kaplan LM,
Rubino F, Mingrone G, Becerra P, Rodriguez P, Vignolo P, Caplan
J, Rodriguez L, Galvao Neto MP. Endoscopic Duodenal Mucosal
Resurfacing for the Treatment of Type 2 Diabetes: 6-Month
Interim Analysis From the First-in-Human Proof-of-Concept
Study. Diabetes Care. 2016 Dec;39(12):2254-2261. doi: 10.2337/
dc16-0383. Epub 2016 Aug 12. PMID: 27519448.
 Rohit Khanna
Rohit Khanna Uma Mahadevan
Uma Mahadevan