Introduction
Spontaneous rupture of the esophagus is also known as Boerhaave syndrome after the Dutch physician who first described it in 1724.1 Historically, Boerhaave syndrome was considered uniformly fatal.
Boerhaave syndrome is a rare entity and is typically seen in middle-aged men after heavy food or alcohol intake, occurring in about 3 per 1,000,000 patients per year.2
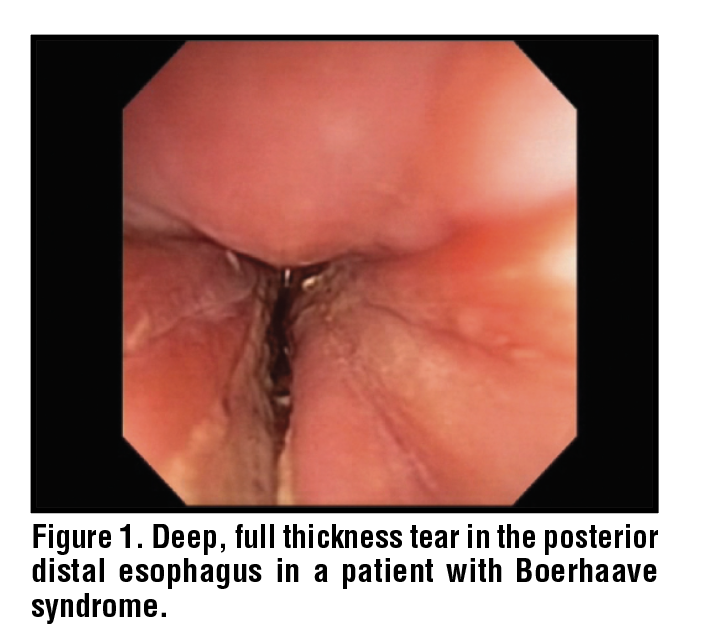
The sudden rise of intraesophageal and intraabdominal pressure against a closed glottis creates a severe pressure gradient across the mediastinum, where the intrathoracic pressure gradient is very low during retching. This, along with dyssynergia between the upper and lower esophageal sphincters, produces a wall stress that results in a full thickness tear – usually occurring along the left posterolateral distal esophagus at the level of the gastroesophageal junction. (Figure 1) Subsequent leakage of luminal contents into the surrounding tissue, if untreated, leads to sepsis, organ failure, and death.3,4
Classically, Boerhaave syndrome presents as Mackler’s triad: vomiting, chest pain, and subcutaneous emphysema.5 One-third of patients present atypically.5 Mortality has historically been high and often ranges from 15% to 42%, but is lower in the modern era.6,7
There has been a paradigm shift in the management of Boerhaave syndrome over the last decade. Earlier, the disorder was treated exclusively with surgery; however, it is now managed with a multidisciplinary approach and thoracic surgical interventions are rarely needed. The overall prognosis is largely determined by prompt diagnosis and treatment. Survival falls dramatically when treatment is delayed beyond 24 hours.8
The Pittsburgh Esophageal Perforation Severity Score (PSS) was originally validated for all causes of esophageal perforation and is a useful tool to stratify risk.9 The score was seen to perform particularly well in the Boerhaave syndrome subgroup (119) in a multinational multicenter retrospective cohort study (1990-2014), done on 288 adults with esophageal perforation.
The PSS assigns points to clinical variables for a maximum score of 18 and classifies patients as low risk (<2), intermediate risk (2–5), or high risk (>5) for death and major complications. Higher PSS values correlate with longer diagnostic delay, favors operative rather than conservative management, predicts increased need for ICU care, prolonged length of hospital stay, and higher in-hospital mortality.10 These findings were corroborated in a retrospective single-center cohort of 56 patients, 21.4% of whom had Boerhaave syndrome.11
Other Esophageal Conditions Predisposing to Boerhaave Syndrome
Boerhaave syndrome may arise in an esophagus that is structurally or functionally abnormal as can be seen in patients with achalasia, eosinophilic esophagitis (EOE), benign strictures, malignancy, and esophageal varices.12 These underlying conditions affect treatment selection and prognosis. In patients with EOE, chronic inflammation leads to remodeling which weakens the esophageal wall and predisposes the patient to transmural rupture.13 On the other hand, high intraluminal pressures along with impaired emptying in patients with achalasia, predispose the esophagus to rupture spontaneously.14
Atypical and High-Risk Presentation in Boerhaave Syndrome
Atypical and high-risk presentations often occur in older or patients with comorbidities. In a retrospective series on 18 hospitalized patients in Turkey, up to one-third of esophageal perforations were initially misdiagnosed as acute coronary syndrome, pulmonary embolism, aortic dissection, perforated peptic ulcer, pancreatitis, pneumonia, or acute hepatitis. This led to delays in definitive treatment and higher mortality.15 A multicenter study in Belarus on 103 hospitalized patients 31 % of the patients were misdiagnosed initially as perforated peptic ulcer, pneumonia, pancreatitis, or acute hepatitis. Several patients underwent unnecessary laparotomy or other procedures.16
Physical Exam/Prognosis and Diagnosis
The majority of patients with Boerhaave syndrome present with chest pain. Other signs may include fever, dyspnea, diaphoresis, and subcutaneous or mediastinal emphysema. Up to one-fifth of cases are initially misdiagnosed.16 Various studies have determined the prognostic factors for Boerhaave syndrome. Older age, significant comorbidities, leukopenia, high CRP, larger tears, thoracic abscess or pleural contamination, overt sepsis, high ASA class and delayed treatment were consistently associated with worse outcomes.18-20
Diagnosis
Retrospective studies support CT-based strategies as first-line imaging. It has a near-100% sensitivity and negative predictive value for ruling out perforation.21,21 CT-esophagography has been shown to be an excellent tool to rule BS in prospective series.23,24 It directly shows contrast leaks and peri-esophageal collections. Water-soluble contrast esophagography remains a useful complementary test, especially to clarify equivocal CT findings.25
Management
Historically Boerhaave syndrome has been managed almost entirely with urgent open surgery (primary repair or esophagectomy plus wide pleuro-mediastinal drainage). Early primary repair was associated with reduced leaks, but was maximally invasive and irreversible.26-28 Modern management has shifted to a multimodal, minimally invasive approach with surgery being reserved for a small minority of cases. In a retrospective cohort study on 80 patients in Germany, patients treated via endoscopic approaches achieved similar survival with less morbidity compared to those who underwent emergency surgery.26
Thoracoscopic/laparoscopic/trans-hiatal approaches are far less frequently employed today, with most patients undergoing endoscopic management as first line therapy.29,30
Stents
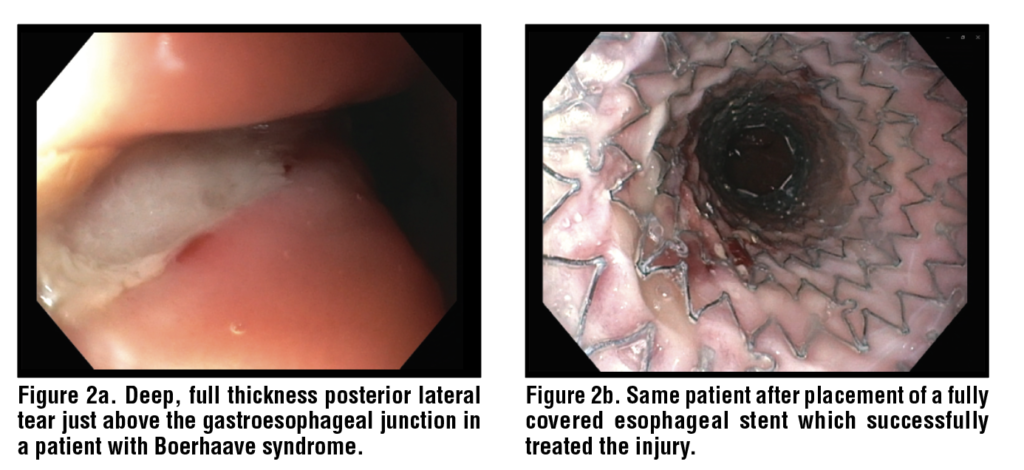
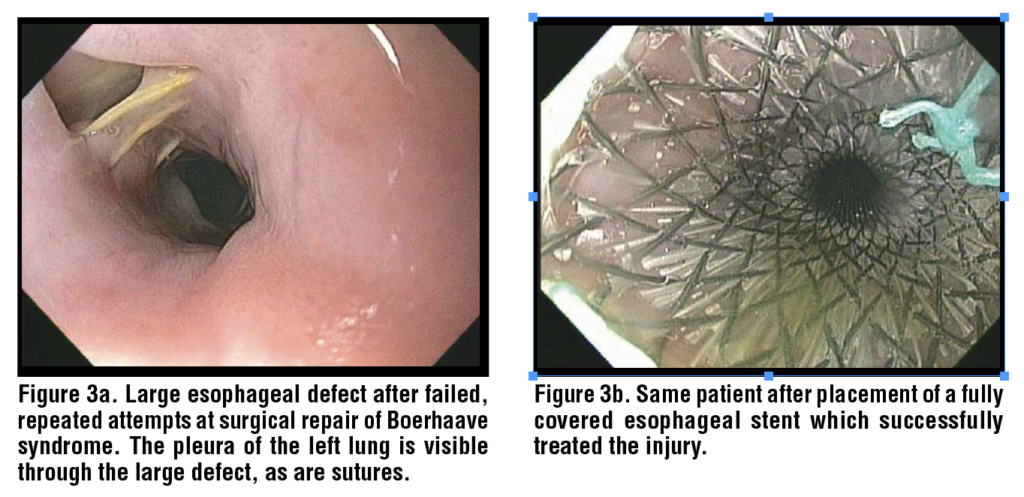
Stents have been used for Boerhaave syndrome dating back to 2001.32 Fully covered and partially covered metal stents as well as plastic esophageal stents have been used (although plastic esophageal stents are now obsolete). (Figures 2 and 3) A systematic review and meta-analysis in 2024 across 18 observational studies on 171 patients with spontaneous esophageal perforations (mostly Boerhaave syndrome patients) reported that closure was achieved in 90/110 patients (82%). In the weighted pooled analysis, the closure rate was 86% (95% CI 77-93%). In the same analysis, failure with stents was reported for 27/160 (17%) with a pooled failure rate of 14% (95% CI 7-22%). Weighted mortality was 6% (95% CI 2–13%).33
In a more recent single center study from Norway that reported on 17 consecutive patients with Boerhaave syndrome (2015-2022), overall outcomes were good. 14 self-expanding metal stents (SEMS) including 12 partially covered and 2 fully covered were utilized. Perforations sealed initially with stent alone in 10, endoscopic vacuum therapy (EVT) alone in 3, and combined stent and EVT in 4 patients. All defects were endoscopically sealed within 12 h of arrival. Mortality was reported only in one patient, however complete endoscopic healing was reported in all surviving patients.
With regards to adverse events, early leakage was reported in 10 patients and 4 patients needed repeat stenting for persistent leaks. Stent migration was reported in 4 patients. Long term adverse events included stricture formation in three patients. Transthoracic drainage via IR was needed in 15/17 patients.34
In a single-center retrospective series from a Dutch tertiary referral center on 21 consecutive Boerhaave syndrome patients, stent-related adverse occurred in 7/19 (37%) stented patients; 3 of these 7 were migrations (two required endoscopic repositioning, one removal).35
Delayed stent migration has been reported as well with a case report of tracheoesophageal fistula forming after multiple stent placements, perforations, diversion, and gastric pull-up for Boerhaave syndrome.36 Another case of small bowel obstruction was seen in a patient with Boerhaave syndrome who was treated with a fully covered esophageal metal stent that had migrated to the jejunum.37
EVT
Endoscopic vacuum therapy (EVT) makes use of continuous negative pressure to drain and collapse an esophageal defect so it can granulate and heal. There are two types of EVT. One is the classic open-pore polyurethane foam, commercially available as Eso-SPONGE / Endo-SPONGE (Boston Scientific, Massachusetts, U.S.A.), the other is an open-pore film sponge (OFD), commercially available as Suprasorb (Lohmann & Rauscher International GmbH & Co. KG, Rengsdorf, Germany). The Endo-SPONGEis mounted on a nasogastric or drainage tube. Then it is placed endoscopically either inside the esophageal lumen across the defect or in the peri-esophageal cavity. A vacuum pump (typically –80 to –150 mmHg) is connected to either of the tubes. It provides constant suction that evacuates pus and secretions, and stimulates granulation and reepithelization. The device needs to be exchanged every 2–4 days until the cavity has collapsed.38 OFD uses a thin open-pore drainage film wrapped around a nasogastric tube, which is often double lumen in design. Its smaller diameter can be passed through narrow strictures or be placed nasally. One lumen is connected to suction and the other can be used for feeding or decompression.39
The first use of EVT to treat patients with Boerhaave syndrome was reported in Germany in 2014. Heits et al. published a single center retrospective series on 10 patients with benign esophageal perforation including 5 Boerhaave syndrome patients who were treated with EVT as the first line therapy. They used the Endo-Sponge system with a of mean 5.4 sessions (2-12) over 19 days. Six patients started EVT within 24 h and four started after 24 h. Delayed initiation of treatment was associated with significantly longer EVT duration and hospital stay. Complete endoscopic healing was seen in 8/10 patients. Failure was reported in two patients with one requirement placement of a covered stent to speed closure. Hospital mortality was 10% though the cause was from cardiovascular collapse after successful EVT. 8/10 patients required pleural drainage either with IR or video-assisted thoracoscopic surgery (VATS). The authors strongly advocated the routine use of CT after placement to look for fluid collections that would warrant additional external drainage.38
Loske et al. used EVT to demonstrate a complete, organ preserving closure in a patient with Boerhaave syndrome. They started with the standard open-pore polyurethane foam drains; then when the perforation had shrunken to a narrow canal, they switched to OFD, allowing continued EVT through the tiny opening.40 A similar case highlighted EVT’s role as a salvage modality after failed primary closure and T-tube drainage.41
A systematic review and meta-analysis analyzing the efficacy and safety of EVT for esophageal luminal defects, including post-surgical anastomotic leaks and transmural perforations (spontaneous or iatrogenic), included 15 single center retrospective cohort studies. A total of 366 patients were included, of which 17.8% were Boerhaave syndrome patients. Clinical success was defined as complete closure of the esophageal defect and was achieved in 87.95% of all patients (95% CI 84.46–91.05%) for all indications. On subgroup analysis it was 88.89% (95% CI 83.22–93.51%) for full-thickness perforations (spontaneous and iatrogenic). The mean duration of treatment was 16.2 days (95% CI 12.6–19.9), with a mean number of 4.6 (95% CI 3.7–5.5) sponge exchanges per patient at a typical interval of 3.7 days (95% CI 3.4–4.1). The attributable mortality of EVT was 4.2% (95% CI 2.3–6.6). Any EVT-related AE was 12.6% (95% CI 10.3–14.7), stricture/dysphagia on follow-up was 5.5% (95% CI 2.1–7.8) and mean sponge migration was 2.6 episodes per patient. 8.5% of patients also received adjunctive SEMS placement in combination with EVT.42
A 2025 retrospective study was performed to assess the efficacy of EVT in a dedicated cohort of Boerhaave syndrome. The retrospective multicenter German study of 57 patients with Boerhaave syndrome treated their patients with either EVT (25/57), stents (SEMS) (15/57) or surgery (14/57). The authors used standard intraluminal or intracavitary sponge/open pore drains with negative pressure. The median duration of treatment with EVT was 17 days (1-84) with a median cycle of 3 exchanges (0-21). Primary EVT success was achieved in 80% of patients, whereas in the non EVT group success was 43.8% and success for those treated with stents only 26.7%. On multivariable analysis, primary EVT was independently associated with treatment success. EVT failure was documented in 5/25 (20%), two patients died during EVT and three required stents or surgery. In-hospital mortality was 8% with EVT, 14% with surgery, and 33% with stenting. EVT should be concomitantly paired with pleural drainage. Forty-two percent of patients underwent percutaneous drainage or VATS, highlighting the fact that EVT often needs to be performed alongside other very invasive interventions.43
True head-to-head studies comparing the different endoscopic modalities specifically in Boerhaave syndrome are lacking, likely due to the emergent nature of the disorder. The largest comparative data come from a single center retrospective German study on 71 esophageal leaks, which included only three patients with Boerhaave syndrome. They treated their patients with either SEMS or EVT. In this study EVT was preferentially used for larger defects (> 9 mm in size) whereas the stented group had leaks of smaller size. The median duration of therapy was 23 days for the EVT group and 33 days for the stented group. EVT achieved a significantly higher leak closure rate (84.4% vs 53.8%, p < 0.05). Adverse events were higher in the stented group with 28% developing strictures and 15% experiencing stent migration. The EVT group had significantly fewer strictures (9.4%) and a low sponge dislocation rate (2.3%).44 EVT therapy, it should be noted, is very cost and labor intensive requiring multiple procedures per patient in a short period of time, which not all patients can tolerate.
VAC Stents
The main underlying challenge in the management of Boerhaave syndrome is prevention and treatment of mediastinal sepsis. Stents are often effective if placed very soon after the injury occurs and before any appreciable fluid collection has developed. Patients with fluid collections may need drains regardless of what other therapies are applied. Negative-pressure endoscopic therapies provide continuous drainage, healing and allow for endoscopic re-assessment.45 Vacuum-assisted stent systems (VACStent GmbH., Germany) combine the benefits of covered stents and EVT by maintaining luminal patency and encouraging early oral intake. VACStent consists of a fully covered self-expanding nitinol stent (similar size to a standard FCSEMS) with a black open-pore polyurethane foam in the mid-portion of the stent. A thin drainage tube exits proximally from the stent-sponge complex to connect to an external vacuum pump. Continuous negative pressure (-80 to -125 mm Hg) pulls the esophageal wall and leak cavity firmly against the sponge sleeve. The combined mechanism allows sealing of the defect with luminal diversion and continuous drainage of the periesophageal cavity. The negative pressure draws the wall snugly on to the sponge, which helps anchor the device and lowers the risk of migration compared with a standard fully covered stent.46
A systematic review on 65 patients with 10 Boerhaave syndrome used VAC Stents for treating esophageal and GEJ transmural defects. Most patients needed a total of 1-3 VAC Stents. The mean duration of therapy was 8.8 +-8.3 days (5 to 14 days) and the average interval for changing the VAC Stent was 5.3 days (2-8). Shorter duration and fewer exchanges may improve patient comfort. Clinical success was noted in 50/65 patients (77%). 23% needed additional intervention mostly with further EVT or surgery.47
Similar outcomes were reported in another retrospective case series on the use of VAC Stents in seven patients, of which one had Boerhaave syndrome. The patient required threeVAC Stents, withsuccessful closure after 21 days.48 Although early data suggest VAC Stent is effective, with shorter treatment, fewer exchanges, and earlier oral intake, robust comparative data with standard EVT is still lacking.
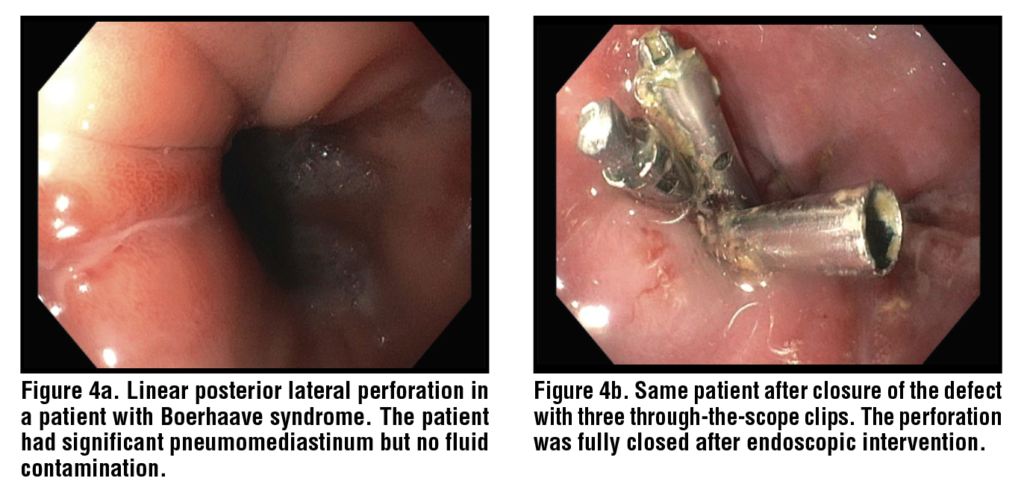
TTS (through-the-scope) Clips and Over the Scope Clips (OTSC)
Clips in the context of Boerhaave syndrome have been used for many years. (Figure 4) They have a clinical efficacy of more than 90% in small (<1–1.5 cm), sharply demarcated perforations with minimal contamination.49 The defects in patients with Boerhaave syndrome are often large with edematous, and may have necrotic ragged edges making them difficult to be approximated by OTSC.
OTSC and TTS have been reported in case series of early contained Boerhaave syndrome or as an adjunct modality.50,51
A case report described how a 2.5 cm Boerhaave syndrome tear was successfully sealed by combining an OTSC with a fully covered self-expanding metal stent. The stent was endoscopically sutured in place to prevent migration. These modalities were combined with aggressive thoracic drainage.52
In a single center series on 14 Boerhaave syndrome patients, 10 were treated primarily with surgery and two with endoscopic therapy and the others with conservative approaches. Overall, 7/14 required SEMS and 3/14 needed OTSC as an adjunct together with thoracic drainage. Primary surgery alone achieved a clinical success rate of 50% whereas endoscopic therapy had 85% clinical success. No patient needed additional surgery and only one death was reported after endoscopy.53
In a case report, a recurrent fibrotic Boerhaave syndrome tear (third transmural rupture) was successfully treated with OTSC and SEMS.54
TTS to treat Boerhaave syndrome has been described only in a handful of case reports. A case series described three patients with contained perforations and these were successfully treated with immediate clipping at presentation. However, the authors advised their use only for short linear defects.55
Conclusion
Outcomes in patients with Boerhaave syndrome are mostly driven by early diagnosis with prompt control of mediastinal sepsis. With the recent advances in therapeutic endoscopy, management has shifted more towards minimally invasive approaches via endoscopy with possible percutaneous catheter drainage, while surgery is needed in only a minority of cases.
No single approach is ideal for every patient. Combined approaches are often required, especially in patients with complex injuries. Observational cohorts have shown that EVT is an emerging option especially for large, contaminated leaks. It has higher closure rates and fewer strictures compared to conventional stenting alone and can be combined with stents as needed. SEMS and VAC Stent systems permit early oral intake with a shorter treatment course. OTSC and TTS clips are often used in patients with early, well-localized perforations with minimal contamination.
References
1. Derbes VJ, Mitchell RE, Jr. Hermann Boerhaave’s Atrocis, nec descripti prius, morbi historia, the first translation of the classic case report of rupture of the esophagus, with annotations. Bull Med Libr Assoc. 1955;43(2):217-40.
2. Horrocks R. Boerhaave syndrome, a rare oesophageal rupture: a case report. Br Paramed J. 2021;5(4):49-53.
3. Connelly CL, Lamb PJ, Paterson-Brown S. Outcomes following Boerhaave’s syndrome. Ann R Coll Surg Engl. 2013;95(8):557-60.
4. Sulpice L, Dileon S, Rayar M, Badic B, Boudjema K, Bail JP, et al. Conservative surgical management of Boerhaave’s syndrome: experience of two tertiary referral centers. Int J Surg. 2013;11(1):64-7.
5. Han D, Huang Z, Xiang J, Li H, Hang J. The Role of Operation in the Treatment of Boerhaave’s Syndrome. Biomed Res Int. 2018;2018:8483401.
6. Biancari F, Tauriainen T, Ylikotila T, Kokkonen M, Rintala J, Mäkäräinen-Uhlbäck E, et al. Outcome of stent grafting for esophageal perforations: single-center experience. Surg Endosc. 2017;31(9):3696-702.
7. Pan J GY, Feng T, Zheng C, Zhang X, Feng S, Sun T, Zhao F, Sha Z, Zhang H. Outcome of treatment modalities for spontaneous esophageal rupture: a meta-analysis and case series. Int J Surg. 2025 Jan 1;111(1):1135-1143. doi: 10.1097/JS9.0000000000001853. PMID: 39051903; PMCID: PMC11745620.
8. Cameron JL, Kieffer RF, Hendrix TR, Mehigan DG, Baker RR. Selective nonoperative management of contained intrathoracic esophageal disruptions. Ann Thorac Surg. 1979;27(5):404-8.
9. Schweigert M, Sousa HS, Solymosi N, Yankulov A, Fernández MJ, Beattie R, et al. Spotlight on esophageal perforation: A multinational study using the Pittsburgh esophageal perforation severity scoring system. J Thorac Cardiovasc Surg. 2016;151(4):1002-9.
10. Abbas G, Schuchert MJ, Pettiford BL, Pennathur A, Landreneau J, Landreneau J, et al. Contemporaneous management of esophageal perforation. Surgery. 2009;146(4):749-55; discussion 55-6.
11. Kovács B, Masuda T, Bremner RM, Smith MA, Huang JL, Hashimi AS, et al. Esophageal perforation: a retrospective report of outcomes at a single center. Annals of Esophagus. 2020;4.
12. Jougon J, Mc Bride T, Delcambre F, Minniti A, Velly JF. Primary esophageal repair for Boerhaave’s syndrome whatever the free interval between perforation and treatment. Eur J Cardiothorac Surg. 2004;25(4):475-9.
13. Kochar T, Dhingra PS, Khaliq MF, McJunkin B. Eosinophilic esophagitis presenting with spontaneous esophageal rupture: a case report. J Med Case Rep. 2019;13(1):275.
14. Moriarity AR, Larkin JO, O’Sullivan KE, Ravi N, Reynolds JV. Spontaneous perforation of the esophagus in a patient with achalasia. Ann Thorac Surg. 2013;96(4):1456-7.
15. Çarkıt S, İpekten F, Karaağaç M, Gök M, Akyuz M. Esophageal perforation management: a single-center experience. Ulus Travma Acil Cerrahi Derg. 2024;30(12):875-82.
16. Panko S KA, Shestjuk A, et al. Diagnosis, management and outcomes of thoracic esophageal perforation. Medical Studies/Studia Medyczne. 2014;30(4):234-240. doi:10.5114/ms.2014.47921.
17. Experience” EPMAS-C, written by Ramzi Addas JB, Claire Renaud, Pierre Berthoumieu, Marcel Dahan, Laurent Brouchet,, Surgery pbOJoT.
18. Sohda M, Kuwano H, Sakai M, Miyazaki T, Kakeji Y, Toh Y, et al. A national survey on esophageal perforation: study of cases at accredited institutions by the Japanese Esophagus Society. Esophagus. 2020;17(3):230-8.
19. Shahriarirad R, Karoobi M, Shekouhi R, Ebrahimi K, Ranjbar K, Amirian A, et al. Esophageal perforation etiology, outcome, and the role of surgical management – an 18-year experience of surgical cases in a referral center. BMC Surg. 2023;23(1):177.
20. Kim JD. Prognostic factors of esophageal perforation and rupture leading to mortality: a retrospective study. J Cardiothorac Surg. 2021;16(1):291.
21. Wei CJ, Levenson RB, Lee KS. Diagnostic Utility of CT and Fluoroscopic Esophagography for Suspected Esophageal Perforation in the Emergency Department. AJR Am J Roentgenol. 2020;215(3):631-8.
22. Awais M, Qamar S, Rehman A, Baloch NU, Shafqat G. Accuracy of CT chest without oral contrast for ruling out esophageal perforation using fluoroscopic esophagography as reference standard: a retrospective study. Eur J Trauma Emerg Surg. 2019;45(3):517-25.
23. Suarez-Poveda T, Morales-Uribe CH, Sanabria A, Llano-Sánchez A, Valencia-Delgado AM, Rivera-Velázquez LF, et al. Diagnostic performance of CT esophagography in patients with suspected esophageal rupture. Emerg Radiol. 2014;21(5):505-10.
24. Evans BA, Craig WY, Cinelli CM, Siegel SG. CT esophagogram in the emergency setting: typical findings and suggested workflow. Emerg Radiol. 2024;31(1):33-44.
25. Madsen HJ, Stuart CM, Wojcik BM, Dyas AR, Hunt A, Helmkamp LJ, et al. Esophagram should be performed to diagnose esophageal perforation before inter-hospital transfer. J Thorac Dis. 2023;15(6):2984-96.
26. Zimmermann M, Hoffmann M, Jungbluth T, Bruch HP, Keck T, Schloericke E. Predictors of Morbidity and Mortality in Esophageal Perforation: Retrospective Study of 80 Patients. Scand J Surg. 2017;106(2):126-32.
27. Axtell AL, Gaissert HA, Morse CR, Premkumar A, Schumacher L, Muniappan A, et al. Management and outcomes of esophageal perforation. Dis Esophagus. 2022;35(1).
28. Allaway MGR, Morris PD, JL BS, Richardson AJ, Johnston ES, Hollands MJ. Management of Boerhaave syndrome in Australasia: a retrospective case series and systematic review of the Australasian literature. ANZ J Surg. 2021;91(7-8):1376-84.
29. Haveman JW, Nieuwenhuijs VB, Kobold JP, van Dam GM, Plukker JT, Hofker HS. Adequate debridement and drainage of the mediastinum using open thoracotomy or video-assisted thoracoscopic surgery for Boerhaave’s syndrome. Surg Endosc. 2011;25(8):2492-7.
30. Lee AHH, Kweh BTS, Gillespie C, Johnson MA. Trans-hiatal repair for Oesophageal and Junctional perforation: a case series. BMC Surg. 2020;20(1):41.
31. Cho JS, Kim YD, Kim JW, I HS, Kim MS. Thoracoscopic primary esophageal repair in patients with Boerhaave’s syndrome. Ann Thorac Surg. 2011;91(5):1552-5.
32. Chung MG, Kang DH, Park DK, Park JJ, Park HC, Kim JH. Successful treatment of Boerhaave’s syndrome with endoscopic insertion of a self-expandable metallic stent: report of three cases and a review of the literature. Endoscopy. 2001;33(10):894-7.
33. Margaris I, Triantafyllou T, Sidiropoulos TA, Sideris G, Theodorou D, Arkadopoulos N, et al. Efficacy of esophageal stents as a primary therapeutic option in spontaneous esophageal perforations: a systematic review and meta-analysis of observational studies. Ann Gastroenterol. 2024;37(2):156-71.
34. Anundsen TK, Førland DT, Johannessen HO, Johnson E. Outcome after stent and endoscopic vacuum therapy-based treatment for postemetic esophageal rupture. Scand J Gastroenterol. 2024;59(1):1-6.
35. Kooij CD, Boptsi E, Weusten B, de Vries DR, Ruurda JP, van Hillegersberg R. Treatment of Boerhaave syndrome: experience from a tertiary center. Surg Endosc. 2025;39(4):2228-38.
36. https://www.atsjournals.org/doi/abs/10.1164/ajrccm.2025.211.Abstracts.A6206.
37. Sain S, Panara C, Jena SS, Yadav A, Nundy S. Small bowel obstruction due to migrated oesophageal metal stent. Int J Surg Case Rep. 2025;128:111034.
38. Heits N, Stapel L, Reichert B, Schafmayer C, Schniewind B, Becker T, et al. Endoscopic endoluminal vacuum therapy in esophageal perforation. Ann Thorac Surg. 2014;97(3):1029-35.
39. Loske G, Albers K, Mueller CT. Endoscopic negative pressure therapy (ENPT) of a spontaneous oesophageal rupture (Boerhaave’s syndrome) with peritonitis – a new treatment option. Innov Surg Sci. 2021;6(2):81-6.
40. E410-E411 ES, 10.1055/s-0034-1392597 D.
41. Kim YE, Do YW, Cho JY, Kim ES, Lee DH. Successful Treatment Using Endoluminal Vacuum Therapy after Failure of Primary Closure in Boerhaave Syndrome. Korean J Gastroenterol. 2019;73(4):219-24.
42. Vohra I, Gopakumar H, Sharma NR, Puli SR. Efficacy of endoscopic vacuum therapy in esophageal luminal defects: a systematic review and meta-analysis. Clin Endosc. 2025;58(1):53-62.
43. Wannhoff A, Kouladouros K, Koschny R, Walter B, Zoll Z, Büringer K, et al. Endoscopic vacuum therapy for the treatment of Boerhaave syndrome: a multicenter analysis. Gastrointest Endosc. 2025;101(2):365-74.
44. Brangewitz M, Voigtländer T, Helfritz FA, Lankisch TO, Winkler M, Klempnauer J, et al. Endoscopic closure of esophageal intrathoracic leaks: stent versus endoscopic vacuum-assisted closure, a retrospective analysis. Endoscopy. 2013;45(6):433-8.
45. Saqib M, Iftikhar M, Ahmed K, Shahid H, Shehr I, Khan Y, et al. Esophageal stenting and endoscopic vacuum therapy for esophageal defects: a systematic review and meta-analysis of observational studies. Ann Med Surg (Lond). 2025;87(9):5963-72.
46. Klose MA, Walldorf J, Damm M, Krug S, Klose J, Ronellenfitsch U, et al. Treatment of esophageal leakages with the Microtech(®)-VAC-Stent: a monocentric early experience of three cases. Ther Adv Gastrointest Endosc. 2023;16:26317745231200312.
47. Kehagias D, Abogabal S, Lampropoulos C, Haider MI, Kehagias I, Jain P, et al. VacStent as a novel therapeutic approach for esophageal perforations and anastomotic leaks- a systematic review of the literature. BMC Surg. 2025;25(1):309.
48. González Aldama M, Santa Cruz Carrera M, López García ML, Prado Troya NF, Irusta Olano L, Jiménez Pérez MA, et al. VACStent therapy for treatment of esophageal transmural defects: early experience in a tertiary hospital. Endoscopy. 2025;57(S 02):eP440.
49. Voermans RP, Le Moine O, von Renteln D, Ponchon T, Giovannini M, Bruno M, et al. Efficacy of endoscopic closure of acute perforations of the gastrointestinal tract. Clin Gastroenterol Hepatol. 2012;10(6):603-8.
50. Bona D, Aiolfi A, Rausa E, Bonavina L. Management of Boerhaave’s syndrome with an over-the-scope clip. Eur J Cardiothorac Surg. 2014;45(4):752-4.
51. Kobara H, Mori H, Rafiq K, Fujihara S, Nishiyama N, Kato K, et al. Successful endoscopic treatment of Boerhaave syndrome using an over-the-scope clip. Endoscopy. 2014;46 Suppl 1 UCTN:E82-3.
52. González-Haba M, Ferguson MK, Gelrud A. Spontaneous esophageal perforation (Boerhaave syndrome) successfully treated with an over-the-scope clip and fully covered metal stent. Gastrointest Endosc. 2016;83(3):650.
53. Tellechea JI, Gonzalez JM, Miranda-García P, Culetto A, D’Journo XB, Thomas PA, et al. Role of Endoscopy in the Management of Boerhaave Syndrome. Clin Endosc. 2018;51(2):186-91.
54. Barakat MT, Girotra M, Banerjee S. (Re)building the Wall: Recurrent Boerhaave Syndrome Managed by Over-the-Scope Clip and Covered Metallic Stent Placement. Dig Dis Sci. 2018;63(5):1139-42.
55. Otsuka K, Murakami M, Ariyoshi T, Yamashita T, Goto S, Watanabe M, et al. Endoscopic clipping of spontaneous esophageal rupture: Case reports of three patients. Int J Surg Case Rep. 2017;38:18-22.
 Taranika Sarkar Das
Taranika Sarkar Das Douglas G. Adler
Douglas G. Adler